8384. Satraplatin
Nomenclature
CAS number: 129580-63-8
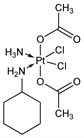
(OC-6-43)-Bis(acetato-κO)amminedichloro(cyclohexanamine)-platinum; bis-acetato-ammine-dichloro-cyclohexylamine-platinum(IV); BMS-182751; BMY-45594; JM-216; Orplatna (GPC). C10H22Cl2N2O4Pt; mol wt 500.28.
C 24.01%, H 4.43%, Cl 14.17%, N 5.60%, O 12.79%, Pt 38.99%.
Description and references
Orally active analog of cisplatin, q.v. Prepn: M. J. Abrams et al., EP 328274; eidem, US 5072011 (1989, 1991 both to Johnson Matthey). Clinical pharmacokinetics: S. Vouillamoz-Lorenz et al., Anticancer Res. 23, 2757 (2003). Clinical evaluation in small-cell lung cancer: E. Fokkema et al., J. Clin. Oncol. 17, 3822 (1999); in hormone-refractory prostate cancer: C. N. Sternberg et al., Oncology 68, 2 (2005). Review of clinical development: L. R. Kelland, Expert Opin. Invest. Drugs 9, 1373-1382 (2000).
Properties
Soly (mg/ml): water ≈0.3; saline 0.4; 1-octanol 0.7. Partition coefficient (octanol/water): 0.1. LD50 in mice (mg/kg): 30 i.p.; 330 orally (Kelland).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Platinum Complexes