8447. Sempervirine
Nomenclature
CAS number: 6882-99-1
2,3,4,13-Tetrahydro-1H-benz[g]indolo[2,3-a]quinolizin-6-ium; 3,4,5,6,14,15,20,21-octadehydroyohimbanium; sempervirene. C19H16N2; mol wt 272.34.
C 83.79%, H 5.92%, N 10.29%.
Description and references
Found in rhizome and roots of Carolina jessamine (yellow jessamine) Gelsemium sempervirens (L.) Ait. f., Loganiaceae: Stevenson, Sayre, J. Am. Pharm. Assoc. 4, 60 (1915); 8, 708 (1919); Hasenfratz, Bull. Soc. Chim. Fr. [4] 53, 1084 (1933); Forsyth et al., J. Chem. Soc. 1945, 579; Prelog, Helv. Chim. Acta 31, 588 (1948). Believed to exist primarily as inner salt depicted below: Woodward, Witkop, J. Am. Chem. Soc. 71, 379 (1949). Synthesis: Woodward, McLamore, ibid. 379; Swan, J. Chem. Soc. 1958, 2039; Ban, Seo, Tetrahedron 16, 11 (1961); Potts, Mattingly, J. Org. Chem. 33, 3985 (1968).
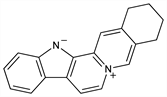
Derivative
Monohydrate.Properties
Yellow needles from chloroform, mp 228°. Brownish-yellow leaflets from dil ethanol, mp 258°. uv max (ethanol): 243, 249, 297, 345, 387 nm (log ε 4.58, 4.57, 4.20, 4.26, 4.24). Dipole moment: 8.5 (dioxane); 7.5 (benzene). Soluble in alcohol, chloroform, pyridine. Slightly sol in acetone. Practically insol in ether, benzene.Derivative
Methochloride.C20H19ClN2; mol wt 322.83.
C 74.41%, H 5.93%, Cl 10.98%, N 8.68%.