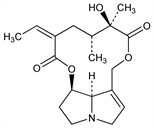
8451. Senecionine
Nomenclature
Description and references
Hepatotoxic pyrrolizidine alkaloid from whole plant of Senecio vulgaris L., Compositae and several other Senecio spp. Isoln: Barger, Blackie, J. Chem. Soc. 1936, 743. Structure: Adams, Govindachari, J. Am. Chem. Soc. 71, 1953 (1949); Koekmoer, Warren, J. Chem. Soc. 1955, 63. Conformation: Culvenor, Tetrahedron Lett. 1966, 1091. Toxicity study: P. N. Harris et al., J. Pharmacol. Exp. Ther. 75, 69 (1942). Review of toxicology of senecionine and other pyrrolizidine alkaloids: E. K. McLean, Pharmacol. Rev. 22, 429-483 (1970). Comprehensive reviews of pyrrolizidine alkaloids: L. Bull et al., The Pyrrolizidine Alkaloids (North-Holland, Amsterdam, 1968) 293 pp; F. L. Warren in The Alkaloids vol. 12, R. H. F. Manske, Ed. (Academic Press, New York, 1970) pp 245-331; D. J. Robins, Fortschr. Chem. Org. Naturst. 41, 115-203 (1982).
Properties
Bitter plates, mp 236°. [α]D25 -55.1° (c = 0.034 in chloroform). Practically insol in water. Freely sol in chloroform; slightly sol in alcohol, ether. LD50 i.v. in mice: 64.12 ±2.24 mg/kg (Harris).Derivative
C15-trans Isomer.Nomenclature
Integerrimine; squalidine; [15(20)E]-12-hydroxysenecionan-11,16,dione.Description and references
Isolated from several Senecio and Crotalaria spp.: L. Bull et al., loc. cit.; F. L. Warren, loc. cit. Identity of squalidine and integerrimine: A. G. Gonzalez, A. Calero, Chem. Ind. (London) 1958, 126.