8464. Serpentine (Alkaloid)
Nomenclature
CAS number: 18786-24-8
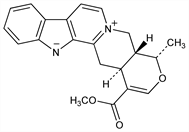
3,4,5,6,16,17-Hexadehydro-16-(methoxycarbonyl)-19α-methyloxayohimbanium. C21H20N2O3; mol wt 348.40.
C 72.40%, H 5.79%, N 8.04%, O 13.78%.
Description and references
From roots of Rauwolfia serpentina (L.) Benth., Apocynaceae: Schlittler, Schwarz, Helv. Chim. Acta 33, 1463 (1950). Structure: Schlittler et al., ibid. 37, 1912 (1954); Klohs et al., J. Am. Chem. Soc. 76, 1332 (1954); Wenkert, Roychaudhuri, ibid. 80, 1613 (1958). Stereoisomer of alstonine, q.v. Stereochemistry: Fritz, Ann. 655, 148 (1962); Shamma, Richey, J. Am. Chem. Soc. 85, 2507 (1963).
Properties
Solvated, yellow rods or leaflets from abs ethanol, mp 158° (air-dried), mp 175° (after drying at 120° in a high vacuum, but still not entirely solvent-free). Dec and turns red on drying at 150°. [α]D25 +292° (c = 0.27 in methanol); [α]D25 +267° (c = 0.21 in ethanol). uv max (ethanol): 252, 308, 370 nm (log ε 4.49, 4.30, 3.61). Freely sol in methanol. Sol in 10% acetic acid, in other organic solvents.Derivative
Nitrate.C21H20N2O3.HNO3; mol wt 411.41.
C 61.31%, H 5.14%, N 10.21%, O 23.33%.
Properties
Yellow crystals, mp 170-172°. Only slightly sol in water.Derivative
Hydrochloride monohydrate.C21H20N2O3.HCl.H2O; mol wt 402.87.
C 62.61%, H 5.75%, N 6.95%, O 15.89%, Cl 8.80%.
Properties
Yellow crystals, mp 246-248°. [α]D23 +178°. Sol in water.Derivative
Perchlorate.C21H20N2O3.HClO4; mol wt 448.85.
C 56.19%, H 4.72%, N 6.24%, O 24.95%, Cl 7.90%.