8486. Siccanin
Nomenclature
Description and references
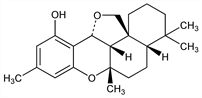
Antibiotic obtained from the cultured broth of Helminthosporium siccans Drechsler, a parasitic organism of rye grass (Lolium multiflorum Lam). Isoln: K. Ishibashi, JP 62 3548 (1962 to Sankyo); idem, J. Antibiot. 15A, 161 (1962). The cis formation of the decahydronaphthalene system may be the first example of the naturally occurring drimane skeleton. Structure: K. Hirai et al., Tetrahedron Lett. 1967, 2177; K. Hirai et al., Tetrahedron 27, 6057 (1971). Approach to synthesis: H. Oida et al., Chem. Pharm. Bull. 20, 2634 (1972); T. Yoshida et al., ibid. 2642. Total synthesis of racemate: M. Kato et al., J. Am. Chem. Soc. 103, 2434 (1981); eidem, Tetrahedron 43, 711 (1987). CD spectral study: S. Nozoe et al., Tetrahedron 30, 2773 (1974). In vitro activity studies: M. Arai et al., Antimicrob. Agents Chemother. 1969, 247; M. G. Bellotti, L. Riviera, Chemioterapia 4, 431 (1985); in vivo studies: S. Sugarwara, Antimicrob. Agents Chemother. 1969, 253. Toxicology: Y. Suzuki et al., Sankyo Kenkyusho Nempo 21, 120 (1969), C.A. 72, 130939k (1970). Mode of action: K. Nose, A. Endo, J. Bacteriol. 105, 176 (1971). Biosynthesis: K. Suzuki et al., Bioorg. Chem. 3, 72 (1974). Clinical evaluation in surface mycosis: D. Crippa et al., G. Ital. Dermatol. Venereol. 120, 57 (1985).
Properties
White to pale yellow odorless crystals, mp 139-140°. [α]D20 -136° (c = 2 in chloroform). uv max (ethanol): 210, 285 nm (ε 45690, 1717). pK 10.9. Very sol in chloroform, dimethylformamide, benzene; freely sol in acetone, ether, ethyl acetate; sol in alcohol. Practically insol in water. LD50 in mice, rats (mg/kg): >6000, >1000 orally; >3000, >600 s.c. or i.p. (Suzuki).Therapeutic Category
Antifungal.
Keywords
Antifungal (Antibiotics)