Nomenclature
Silibin; silibinin; silybum substance E6; silymarin I. Description and references
Hepatoprotective flavonoid compounds isolated
from the fruit or seeds of milk thistle, Silybum marianum L. Gaertn. (Asteraceae); major constituent of the therapeutic
extract, silymarin. Exists as a mixture of diastereomers A and B.
Isoln and chemistry: H. Wagner et al., Arzneim.-Forsch. 18, 688 (1968); 24, 466 (1974). Structural studies: R. H"ansel, G. Sch"opflin, Tetrahedron Lett. 8, 3645 (1967);
A. Pelter, R. H"ansel, ibid. 9, 2911 (1968);
R. H"ansel et al., J. Chem. Soc.
Chem. Commun. 1972, 195. Biomimetic synthesis:
L. Merlini et al., ibid. 1979, 695; eidem, J. Chem. Soc. Perkin Trans. 1 1980, 775.
Pharmacology and toxicology: G. Hahn et al., Arzneim.-Forsch. 18, 698 (1968).
Structure and stereochemistry of silybin and isosilybin diastereomers:
D. Y.-W. Lee, Y. Liu, J. Nat. Prod. 66, 1171 (2003); N.-C. Kim et al., Org. Biomol. Chem. 1, 1684 (2003).
HPLC determn in plasma: C. S. L. Hoh et al., J. Agric. Food Chem. 55, 2532
(2007); and clinical pharmacokinetics of complex with phosphatidylcholine:
W. Li et al., J. Chromatogr.
B 862, 51 (2008). Mechanism of antioxidant
action: P. Trouillas et al., J. Phys. Chem. A 112, 1054 (2008). Review
of mechanism of action and therapeutic potential: R. Gazak et
al., Curr. Med. Chem. 14, 315-338 (2007).
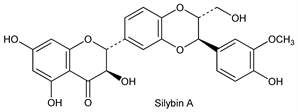
Derivative
Silybin A.
Nomenclature
CAS number: 22888-70-6
(2
R,3
R)-2-[(2
R,3
R)-2,3-Dihydro-3-(4-hydroxy-3-methoxyphenyl)-2-(hydroxymethyl)-1,4-benzodioxin-6-yl]-2,3-dihydro-3,5,7-trihydroxy-4
H-1-benzopyran-4-one.
C
25H
22O
10; mol wt 482.44.
C 62.24%, H 4.60%, O 33.16%.
Properties
Yellowish flat crystals from CH3OH
+ H2O, mp 162-163°. [α]D +20° (c = 0.21 in acetone) (Lee, Liu). Also reported
as mp 158-160°.
[α]D +6.1° (c = 0.3 in methanol). uv max (methanol):
217, 230, 288 nm (log ε 5.2, 5.2, 5.1) (Kim). Sol in acetone, ethyl
acetate, methanol, ethanol; sparingly sol in chloroform. Practically
insol in water.Derivative
Silybin B.
Nomenclature
CAS number: 142797-34-0
(2
R,3
R)-2-[(2
S,3
S)-2,3-Dihydro-3-(4-hydroxy-3-methoxyphenyl)-2-(hydroxymethyl)-1,4-benzodioxin-6-yl]-2,3-dihydro-3,5,7-trihydroxy-4
H-1-benzopyran-4-one.
C
25H
22O
10; mol wt 482.44.
C 62.24%, H 4.60%, O 33.16%.
Properties
Yellow grain crystals from CH3OH +
H2O, mp 158-160°. [α]D -1.07° (c = 0.28 in acetone) (Lee, Liu). Also reported
as mp 157-159°.
[α]D +0.6° (c = 0.3 in methanol). uv max (methanol):
217, 230, 288 nm (log ε 5.2, 5.3, 5.2) (Kim).Derivative
Isosilybin A.
Nomenclature
CAS number: 142796-21-2
(2
R,3
R)-2-[(2
R,3
R)-2,3-Dihydro-2-(4-hydroxy-3-methoxyphenyl)-3-(hydroxymethyl)-1,4-benzodioxin-6-yl]-2,3-dihydro-3,5,7-trihydroxy-4
H-1-benzopyran-4-one.
C
25H
22O
10; mol wt 482.44.
C 62.24%, H 4.60%, O 33.16%.
Properties
Colorless needles from CH3OH + H2O, mp 201-203°. [α]D +48.15° (c = 0.27 in acetone) (Lee, Liu). Also reported
as mp 158-160°.
[α]D +30.4° (c = 0.3 in methanol). uv max (methanol):
212, 230, 288 nm (log ε 5.4, 5.3, 5.1) (Kim).Derivative
Isosilybin B.
Nomenclature
CAS number: 142796-22-3
(2
R,3
R)-2-[(2
S,3
S)-2,3-Dihydro-2-(4-hydroxy-3-methoxyphenyl)-3-(hydroxymethyl)-1,4-benzodioxin-6-yl]-2,3-dihydro-3,5,7-trihydroxy-4
H-1-benzopyran-4-one.
C
25H
22O
10; mol wt 482.44.
C 62.24%, H 4.60%, O 33.16%.
Properties
Colorless needles from CH3OH + H2O, mp 236-238°. [α]D -23.55° (c = 0.31 in acetone) (Lee, Liu). Also reported
as mp 194-196°.
[α]D -17.6° (c = 0.3 in methanol). uv max (methanol):
212, 230, 288 nm (log ε 5.4, 5.3, 5.1) (Kim).