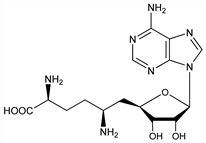
8544. Sinefungin
Nomenclature
Description and references
Adenine-containing antibiotic produced by Streptomyces griseoleus NRRL 3739. Isoln: R. L. Hamill, M. M. Hoehn, US 3758681 (1973 to Lilly); eidem, J. Antibiot. 26, 463 (1973). Antifungal activity: R. S. Gordee, T. F. Butler, ibid. 466. Antiprotozoal activity: D. K. Dube et al., Am. J. Trop. Med. Hyg. 32, 31 (1983); A. Ferrante et al., Trans. R. Soc. Trop. Med. Hyg. 78, 837 (1984); P. Paolantonacci et al., Antimicrob. Agents Chemother. 28, 528 (1985); E. Zweygarth et al., Trop. Med. Parasitol. 37, 255 (1986); in vivo antileishmanial activity: J. L. Avila et al., Am. J. Trop. Med. Hyg. 43, 139 (1990). Synthesis: M. Geze et al., J. Am. Chem. Soc. 105, 7638 (1983); stereoselective synthesis: M. P. Maguire et al., J. Org. Chem. 55, 948 (1990). Inhibition of methyltransferase: R. T. Borchardt et al., Biochem. Biophys. Res. Commun. 89, 919 (1979).