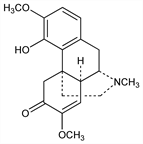
8546. Sinomenine
Nomenclature
Description and references
An optical isomer of methoxythebainone. Configuration at the asymmetric centers, C9, C13, and C14, is the mirror image of those in morphine. From root of Sinomenium acutum (Thunb.) Rehd. & Wils. (Cocculus diversifolius DC.), Menispermaceae: Goto, J. Chem. Soc. Jpn. 44, 795 (1923), C.A. 18, 2710 (1924); Ohta, Kitasato Arch. Exp. Med. 6, 259 (1925), C.A. 21, 32332 (1927). Structure: Kondo, Ochiai, Ann. 470, 224 (1929); Sasaki, Yakugaku Zasshi 80, 270 (1960), C.A. 54, 11702g (1960). Biogenesis: Cohen, Chem. Ind. (London) 1956, 1391. Pharmacology: S. Fu et al., Yao Hsueh Hsueh Pao 10, 673 (1963), C.A. 60, 8525h (1964). Reviews: Holmes in R. H. F. Manske, H. L. Holmes, The Alkaloids vol. II (Academic Press, New York, 1952) pp 219-260; K. W. Bentley, The Chemistry of the Morphine Alkaloids (Oxford, 1954).