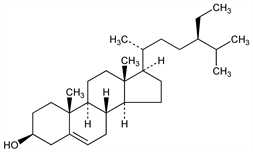
8556. β-Sitosterol
Nomenclature
Description and references
Common sterol in plants. Isoln from wheat germ oil, corn oil: Anderson et al., J. Am. Chem. Soc. 48, 2987 (1926); from rye germ oil: Gloyer, Schuette, ibid. 61, 1901 (1939); from cottonseed oil: Wallis, Chakravorty, J. Org. Chem. 2, 335 (1937); from tall oil: Sandqvist, Bengtsson, Ber. 64, 2167 (1931). Also occurs in soy and calabar beans, in rice embryos: Tanaka, J. Biochem. 17, 483 (1933); in cascara, cinchona bark and cinchona wax: Dirscherl, Z. Physiol. Chem. 235, 1 (1935). Prepn from tall oil: G. I. Fujimoto, A. E. Jacobson, J. Org. Chem. 29, 3377 (1964). Identity with 22:23-dihydrostigmasterol: W. Dirscherl, H. Nahm, Ann. 555, 57 (1944). Identity with cinchol: eidem, Ann. 558, 231 (1947). Structure: Bernstein, Wallis, J. Org. Chem. 2, 341 (1937); Bergmann, Low, ibid. 12, 67 (1947); Shoppee, J. Chem. Soc. 1948, 1032. Stereospecific synthesis: W. Sucrow, M. Slopianka, Ber. 108, 3721 (1975). Clinical efficacy in treatment of type II hyperlipoproteinemia: R. S. Lees, A. M. Lees, in Lipoprotein Metabolism, H. Greten, Ed. (Springer-Verlag, New York, 1976) pp 119-124; P. Oster et al., ibid. pp 125-130. Studies on inhibition of cholesterol absorption: S. M. Grundy, H. Y. I. Mok, ibid. pp 112-118; I. Ikeda, M. Sugano, Biochim. Biophys. Acta 732, 651 (1983). Inhibition of induced carcinogenesis: N. D. Nigro et al., J. Natl. Cancer Inst. 69, 103 (1982). Clinical trial in treatment of prostatic adenoma: H.-P. Szutrely, Med. Klin. 77, 520 (1982). Book: Monographs on Atherosclerosis Vol. 10, T. B. Clarkson et al., Eds. entitled “Sitosterol” by O. J. Pollak, D. Kritchevsky (Karger, Basel, 1981) 219 pp.
Properties
Plates from alcohol, mp 140°. [α]D25 -37° (c = 2 in chloroform).Derivative
Acetate.Properties
mp 127-128°. [α]D25 -41° (c = 2 in chloroform).Therapeutic Category
Antilipemic. Treatment of prostatic adenoma.
Keywords
Antilipemic