8737. Sparteine
Nomenclature
Description and references
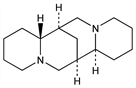
In yellow and black lupin beans, Lupinus luteus L. and L. niger Hort.; also in Cytisus scoparius (L.) Link. and Anagyris foetida L., Leguminosae. Extraction procedure: Karrer et al., Helv. Chim. Acta 11, 1062 (1928). Structure: Clemo et al., J. Chem. Soc. 1931, 429; Ing, ibid. 1933, 504; Schirm, Besendorf, Arch. Pharm. 280, 64 (1942); Galinovsky, Stern, Ber. 77B, 132 (1944); Clemo et al., J. Chem. Soc. 1949, 663. Biosynthesis: Anet et al., Nature 165, 35 (1950); Schpf et al., Angew. Chem. 65, 161 (1953); 69, 69 (1957); van Tamelen, Foltz, J. Am. Chem. Soc. 82, 2400 (1960). Absolute configuration: Okuda et al., Chem. Ind. (London) 1961, 1116. Conformation: Bohlmann et al., Tetrahedron Lett. 1965, 2705; Wiewiorowski et al., Can. J. Chem. 45, 1447 (1967). Synthesis of racemate: Bohlmann et al., Ber. 106, 3026 (1973); N. Takatsu et al., Chem. Pharm. Bull. 35 4990 (1987). Chemistry: Binnig, Arzneim.-Forsch. 24, 752 (1974). Studies of cardiovascular effects: Raschak, ibid. 753.
Properties
Viscous, oily liquid. bp8 173°. Volatile with steam. [α]D21 16.4° (c = 10 in abs alc). nD20 1.5312. d420 1.020. pK1 at 20°: 2.24; pK2: 9.46; pH of 0.01 molar soln 11.6. One gram dissolves in 325 ml water. Freely sol in alcohol, chloroform or ether.Derivative
Sulfate pentahydrate.Nomenclature
Properties
Columnar crystals, loses water of crystn at 100° turning brown, dec 136°. pH of 0.05 molar soln 3.3. One gram dissolves in 1.1 ml water, 3 ml alcohol. Practically insol in chloroform and ether.Derivative
l-α-Isosparteine.Nomenclature
Description and references
14aα-stereoisomer. Isoln from Cytisus scoparius (L.) Link, (Spartium scoparium L.), Leguminosae: Valeur, Compt. Rend. 167, 23, 163 (1918); from Lupinus caudatus Kellog, Leguminosae: Marion et al., Can. J. Chem. 29, 22 (1951). Identity as stereoisomer: Marion, Leonard, ibid. 297. Structure: Leonard et al., J. Am. Chem. Soc. 77, 1552 (1955). Absolute configuration: Okuda, Tsuda, Chem. Ind. (London) 1961, 1115.
Properties
Monohydrate, needles from boiling acetone, mp 108-110°. [α]D22 51.6° (c = 0.7 in abs. ethanol).Therapeutic Category
In treatment of cardiac insufficiency.