8739. Spectinomycin
Nomenclature
Description and references
Antibiotic isolated from fermentation broth of Streptomyces spectabilis: D. J. Mason et al., Antibiot. Chemother. 11, 118 (1961); M. E. Bergy et al., ibid. 661; M. E. Bergy, C. De Boer, US 3234092 (1966 to Upjohn). Purification and crystallization: H. K. Jahnke, US 3206360; V. J. Peters, US 3272706 (1965, 1966 both to Upjohn). Isoln and characterization: A. C. Sinclair, A. F. Winfield, Antimicrob. Agents Chemother. 1961, 503. Structure: H. Hoeksema et al., J. Am. Chem. Soc. 84, 3212 (1962); P. F. Wiley et al., ibid. 85, 2652 (1963). Solubility data: J. R. Marsh, P. J. Weiss, J. Assoc. Off. Anal. Chem. 50, 457 (1967). Stereochemistry and abs config: T. G. Cochran et al., Chem. Commun. 1972, 494. Biosynthesis: L. A. Mitscher et al., ibid. 1971, 1541; H. Otsuka et al., J. Am. Chem. Soc. 102, 6817 (1980). Enantioselective synthesis: D. R. White et al., Tetrahedron Lett. 1979, 2737; S. Hanessian, R. Roy, Can. J. Chem. 63, 163 (1985). Mechanism of action study: M. F. Brink et al., Nucleic Acids Res. 22, 325 (1994). HPLC determn in animal plasma: N. Haagsma et al., J. Chromatogr. 615, 289 (1993). Clinical pharmacokinetics: J. G. Wagner et al., Int. Z. Klin. Pharmakol. Ther. Toxikol. 1, 261 (1968). Clinical evaluation in gonorrhea: Y. H. Kouri et al., Genitourin. Med. 65, 342 (1989); in chancroid: M. Guzmán et al., Sex. Transm. Dis. 19, 291 (1992). Co-treatment with lincomycin, q.v., for footrot in sheep: C. M. Venning et al., Aust. Vet. J. 67, 258 (1990). Reviews: W. M. McCormack, M. Finland, Ann. Intern. Med. 84, 712-716 (1976); W. J. Holloway, Med. Clin. North Am. 66, 169-173 (1982).
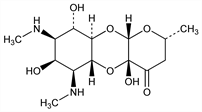
Properties
Amorphous powder. [α]D25 20° (water). pKa′1 6.95, pKa′2 8.70. Sol in water, methanol, ethanol. Insol in acetone, hydrocarbon solvents.Derivative
Dihydrochloride pentahydrate.Nomenclature
Description and references
Exists as a ketone hydrate and not in the carbonyl form (Cochran et al.).
Properties
Colorless needles from aq acetone, mp 205-207° (dec). [α]D +14.8° (c = 0.42 in water). Soly in water, methanol, propylene glycol, formamide, DMSO, 0.1N NaOH, 0.1N HCl: >20 mg/ml. Soly (mg/ml): chloroform 0.042; acetone 0.015; diethyl ether 0.010.Derivative
Sulfate tetrahydrate.Nomenclature
Properties
White crystals from aq acetone, mp ≈185° (dec). [α]D25 +17.0° (c = 1 in water). pKa′1 7.00, pKa′2 8.75. Soly in water, DMSO: 225, 5-10 (mg/ml). Practically insol in pyridine, chloroform, ethanol, ethyl acetate, cyclohexane, benzene, acetone, DMF, dioxane, acetonitrile.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Aminoglycosides