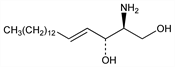
8747. Sphingosine
Nomenclature
Description and references
Important membrane component; long-chain amino-dialcohol; moiety of certain phosphatides, such as sphingomyelins, cerebrosides, and gangliosides. Natural sphingosine is d-(+)-erythro-1,3-dihydroxy-2-amino-4-trans-octadecene. Does not exist in the free state in animals, plants or microorganisms. First obtained by hydrolysis of cerebrosides from brains: J. L. W. Thudichum, Die Konstitution des Gehirns des Menschen und der Tiere (Tübingen, 1901). Separation procedures: Carter et al., J. Biol. Chem. 170, 269 (1947); Wittenberg, ibid. 216, 379 (1955); Tipton, Biochem. Prep. 9, 127 (1962). Stereospecific synthesis of dl-erythro-trans-form and isomers: C. A. Grob, F. Gadient, Helv. Chim. Acta 40, 1145 (1957). Stereoselective synthesis: H. Newman, J. Am. Chem. Soc. 95, 4098 (1973); B. Bernet, A. Vasella, Tetrahedron Lett. 24, 5491 (1983). Comprehensive review: C. A. Grob, Record Chem. Progr. (Kresge-Hooker Sci. Lib.) 18, 55-66 (1957); D. Shapiro, Chemistry of Sphingolipids (Hermann, Paris, 1969) 111 pp. Bibliography: Rodd's Chemistry of Carbon Compounds Vol. I, part E, S. Coffey, Ed. (Elsevier, New York, 2nd ed., 1976) pp 394-397.