8760. Spironolactone
Nomenclature
Description and references
Aldosterone antagonist. Prepn: Cella, Tweit, J. Org. Chem. 24, 1109 (1959); US 3013012 (1961 to Searle); Tweit et al., J. Org. Chem. 27, 3325 (1962). Activity and metabolic studies: Gerhards, Engelhardt, Arzneim.-Forsch. 13, 972 (1963). Crystal and molecular structure: Dideberg, Dupont, Acta Crystallogr. B28, 3014 (1972). Comprehensive description: J. L. Sutter, E. P. K. Lau, Anal. Profiles Drug Subs. 4, 431-451 (1975). Review of carcinogenetic risk: IARC Monographs 24, 259-273 (1980). Review of antiandrogen effects and clinical use in hirsutism: R. R. Tremblay, Clin. Endocrinol. Metab. 15, 363-371 (1986); of clinical efficacy in hypertension: A. N. Brest, Clin. Ther. 8, 568-585 (1986). Review of pharmacology: H. A. Skluth, J. G. Gums, DICP Ann. Pharmacother. 24, 52-59 (1990). Clinical trial in congestive heart failure: B. Pitt et al., N. Engl. J. Med. 341, 709 (1999).
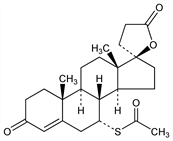
Properties
Crystals from methanol, mp 134-135° (resolidifies and dec 201-202°). [α]D20 33.5° (chloroform). uv max: 238 nm (ε 20200). Practically insol in water. Sol in alcohol; freely sol in benzene, chloroform. LD50 in rats, mice, rabbits (mg/kg): 790, 360, 870 i.p. (IARC, 1980).Therapeutic Category
Diuretic.
Therapeutic Category (Veterinary)
Diuretic.Keywords
Aldosterone Antagonist; Diuretic; Steroids