8771. Stachydrine
Nomenclature
CAS number: 471-87-4
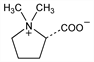
(2S)-2-Carboxy-1,1-dimethylpyrrolidinium inner salt; methyl hygrate betaine; hygric acid methylbetaine. C7H13NO2; mol wt 143.18.
C 58.72%, H 9.15%, N 9.78%, O 22.35%.
Description and references
Occurs widely in nature, especially in alfalfa, chrysanthemum, citrus and stachys species. May be prepd by methylation of proline. Isoln: Planta, Schulze, Ber. 26, 939 (1893); Jahns, ibid. 29, 2065 (1896); Yoshimura, Z. Physiol. Chem. 88, 334 (1913); Vickery, J. Biol. Chem. 61, 117 (1924). Structure: Schulze, Trier, Z. Physiol. Chem. 67, 59 (1910). Synthesis: Karrer, Widmer, Helv. Chim. Acta 8, 364 (1925). Biosynthesis: Robertson, Marion, Can. J. Chem. 38, 396 (1960). Review: Marion in The Alkaloids vol. 1, R. H. F. Manske, H. L. Holmes, Eds. (Academic Press, New York, 1950) pp 101-103.
Derivative
Monohydrate.Properties
Deliquescent crystals, sweetish taste, mp 235° when anhydr. Isomerizes at the mp to methyl hygrate. Sol in water, alcohol, dil acids. Practically insol in ether, chloroform.Derivative
Hydrochloride.C7H13NO2.HCl; mol wt 179.64.
C 46.80%, H 7.86%, N 7.80%, O 17.81%, Cl 19.74%.
Properties
Large prisms from abs alcohol, dec 235°, very sol in water, sol in 13 parts alcohol.Derivative
Acid oxalate.C7H13NO2.C2H2O4; mol wt 233.22.
C 46.35%, H 6.48%, N 6.01%, O 41.16%.
Properties
Needles, mp 106°. Practically insol in abs alcohol.Derivative
Aurichloride.C7H13NO2.HAuCl4; mol wt 482.97.
C 17.41%, H 2.92%, N 2.90%, O 6.63%, Au 40.78%, Cl 29.36%.
Properties
Yellow leaflets, mp 225° (rapid heating). Practically insol in cold water. Quite sol in hot water.Derivative
Platinichloride tetrahydrate.(C7H13NO2)2.H2PtCl6.4H2O; mol wt 768.25.
C 21.89%, H 4.72%, N 3.65%, O 16.66%, Pt 25.39%, Cl 27.69%.