8876. Succinylcholine Iodide
Nomenclature
CAS number: 541-19-5
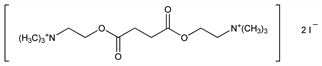
O,O-Succinyldicholine iodide; diacetylcholine iodide; diacetylcholine diiodide; suxamethonium iodide; bis(β-dimethylaminoethyl)succinate bis(methyl iodide); Celocurine (Pfizer). C14H30I2N2O4; mol wt 544.21.
C 30.90%, H 5.56%, I 46.64%, N 5.15%, O 11.76%.
Description and references
Prepd by reacting β-bromoethyl succinate with trimethylamine: Glick, J. Biol. Chem. 137, 357 (1941); Fusco et al., Gazz. Chim. Ital. 79, 129, 837 (1949); Walker, J. Chem. Soc. 1950, 193. Succinic acid chloride may be coupled with choline chloride directly or with dimethylaminoethanol, followed by quaternization with methyl iodide: Tammelin, Acta Chem. Scand. 7, 185 (1953). A third method is to start with the diethyl ester of succinic acid, then bring about the exchange reaction with dimethylaminoethanol, and quaternize with methyl iodide: Phillips, J. Am. Chem. Soc. 71, 3264 (1949).
Properties
Slightly hygroscopic crystals, mp 243-245°. Freely sol in water or normal saline, giving solns which are very slightly acidic. Most stable at pH 4-5. Aq solns undergo progressive hydrolysis with corresp loss of activity and increase in acidity. Prepare solns just before using. Incompatible with solns of alkaline salts.Therapeutic Category
Neuromuscular blocking agent.
Therapeutic Category (Veterinary)
Muscle relaxant (skeletal).Keywords
Neuromuscular Blocking Agent; Depolarizing Agents