8890. Sulbenicillin
Nomenclature
CAS number: 41744-40-5
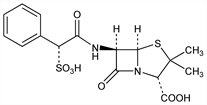
(2S,5R,6R)-3,3-Dimethyl-7-oxo-6-[[(2R)-phenylsulfoacetyl]amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; α-sulfobenzylpenicillin; sulfocillin. C16H18N2O7S2; mol wt 414.45.
C 46.37%, H 4.38%, N 6.76%, O 27.02%, S 15.47%.
Description and references
Semi-synthetic antibiotic related to penicillin. Prepn: S. Morimoto et al., DE 1948943 corresp to US 3660379 (1970, 1972 both to Takeda); eidem, J. Med. Chem. 15, 1105, 1108 (1972). In vitro activity studies: Tsuchiya et al., J. Antibiot. 24, 607 (1971); in vivo: Yamazaki, Tsuchiya, ibid. 620. Metabolism: Tsuchiya et al., ibid. 25, 336 (1972). Toxicity: Y. Murata et al., Takeda Kenkyushoho 30, 262 (1971), C.A. 76, 147x (1972). Pharmacokinetics: A. Montanari et al., Clin. Ter. 89, 163 (1979); eidem, Int. J. Clin. Pharmacol. Ther. Toxicol. 18, 225 (1980).
Derivative
Disodium salt.Nomenclature
Kedacillina (Bracco); Sulpelin (Senju); Lilacillin (Takeda).C16H16N2Na2O7S2; mol wt 458.42.
C 41.92%, H 3.52%, N 6.11%, Na 10.03%, O 24.43%, S 13.99%.
Properties
Prepd as a 3:1 mixture of d() and l(+) isomers. Yellowish-white powder, mp 195-198° (dec). [α]D22 +169-173°. uv max: 257, 262, 268 nm. Very sol in water; sol in methanol. Almost insol in n-propanol, acetone, chloroform, benzene, ethyl acetate. LD50 in male, female mice, male, female rats (mg/kg): 7900, 8000, 6000, 6200 i.v.; 9600, 10000, 7200, 7500 i.p.; 11500, 13500, 11000, 11800 s.c.; 11000, 10500, 8300, 8600 i.m.; all >15000 orally (Murata).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins