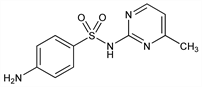
8913. Sulfamerazine
Nomenclature
Description and references
Prepd by condensing 2-amino-4-methylpyrimidine with acetylsulfanilyl chloride followed by hydrolysis of the acetyl group: Roblin et al., J. Am. Chem. Soc. 62, 2002 (1940); Sprague et al., ibid. 63, 3028 (1941); Sprague, US 2407966 (1946 to Sharp & Dohme). For prepn of 2-amino-4-methylpyrimidine see Benary, Ber. 63, 2601 (1930); Backer, Grevenstuk, Rec. Trav. Chim. 61, 291 (1942); cf. E. H. Northey, Sulfonamides (Reinhold, New York, 1948). Antimicrobial activity: Gill et al., Indian J. Vet. Sci. 32, 240 (1962); Vaichulis, Vedros, Chemotherapia 11, 315 (1966). Toxicity studies: Simunek et al., Vet. Med. (Prague) 13, 619 (1968). Kinetics of sulfamerazine decompn: Zajac, Diss. Pharm. Pharmacol. 22, 455 (1970). Comprehensive description: R. D. G. Woolfenden, Anal. Profiles Drug Subs. 6, 515-577 (1977).
Properties
Crystals, mp 234-238°. uv max (water): 243, 257 nm (E1%1cm 875, 822); (0.1M HCl): 243, 307 nm (E1%1cm 625, 200); (ethanol): 271 nm (E1%1cm 835). Slowly darkens on exposure to light. Soly in water at 37°: 35 mg/100 ml at pH 5.5; 170 mg/100 ml at pH 7.5. Readily sol in dil mineral acids and in solns of potassium, ammonium and sodium hydroxides. Sparingly sol in acetone, slightly sol in alcohol, very slightly sol in ether, chloroform.Derivative
Monosodium salt.Nomenclature
Properties
Crystals. Bitter, caustic taste. Hygroscopic. On prolonged exposure to humid air, it absorbs CO2 with the liberation of sulfamerazine and becomes incompletely sol in water. Its solns are alkaline to phenolphthalein (pH 10 or more). One gram dissolves in 3.6 ml water. Slightly sol in alc. Insol in ether, chloroform.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Sulfonamides