8931. Sulfanilylurea
Nomenclature
CAS number: 547-44-4
4-Amino-N-(aminocarbonyl)benzenesulfonamide; N-sulfanilylcarbamide; sulfacarbamide; sulfaurea; Euvernil (Heyden); Uractyl; Uramid (Spofa); Urenil; Urosulfan. C7H9N3O3S; mol wt 215.23.
C 39.06%, H 4.21%, N 19.52%, O 22.30%, S 14.90%.
Description and references
Prepd by treating N4-acetylsulfanilamide with potassium cyanate or with carbamyl chloride or with urea (or with nitrourea and sodium carbonate) in 80% alcohol. The p-AcNHC6H4SO2NHCONH2 is saponified by slight warming with dil KOH and then acidified: Martin et al., US 2411661 (1946 to Geigy). By boiling sulfanilamide with urea and sodium carbonate in 75% alcohol: Haack, Alien Prop. Custodian, Serial 369, 118 (1943). By warming calcium acetylsulfanilylcyanamide with dil HCl: Winnek et al., J. Am. Chem. Soc. 64, 1684 (1942); improved procedure: Leitch et al., Can. J. Res. 23B, 139 (1945).
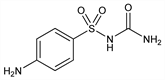
Properties
Crystals from water. mp 146-148° (slight dec). Solubility in water at 37°: 811 mg/100 ml. Soluble in alkalies. Forms a very soluble sodium salt.Derivative
Monohydrate.Properties
mp 125-127°.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Sulfonamides