8946. Sulfazecin
Nomenclature
CAS number: 77912-79-9
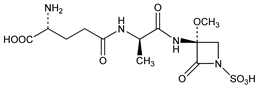
d-γ-Glutamyl-N-[(3R)-3-methoxy-2-oxo-1-sulfo-3-azetidinyl]-d-alaninamide; 3-γ-d-glutamyl-d-alanylamino-3-methoxyazetidin-2-one-1-sulfonic
acid; (3R)-3-(γ-d-glutamyl-d-alanylamino)-3-methoxy-2-oxoazetidine-1-sulfonic
acid; antibiotic G-6302. C12H20N4O9S; mol wt 396.37.
C 36.36%, H 5.09%, N 14.13%, O 36.33%, S 8.09%.
Description and references
Novel monocyclic β-lactam (monobactam) antibiotic, produced by Pseudomonas acidophila and P. mesoacidophila. Isoln, description of physico-chemical and bacteriostatic properties: A. Imada et al., DE 2855949 corresp to US 4229436 (1979, 1980 both to Takeda); eidem, Nature 289, 590 (1981); M. Asai et al., J. Antibiot. 34, 621 (1981). X-ray crystallographic structure determn: K. Kamiya et al., Acta Crystallogr. B37, 1626 (1981). Prepn of fluorinated sulfazecin analogs: K.Yoshioka et al., J. Org. Chem. 49, 1427 (1984); P. F. Bevilacqua et al., ibid. 1430.
Properties
Crystallizes from methanol/water as the alcoholate-hemihydrate; colorless needles, mp 168-170°. [α]D25 +82° (c = 1.0 in water). pKa: 3.4 (COO); 9.2 (NH3+). Sol in water, DMF, DMSO. Slightly sol in methanol, THF. Practically insol in ethanol, acetone, ethyl acetate, chloroform and other organic solvents. Relatively stable in neutral and weakly acidic solns; unstable in alkaline and strongly acidic solns.Derivative
Sodium salt monohydrate.C12H19N4NaO9S.H2O; mol wt 436.37.
C 33.03%, H 4.85%, N 12.84%, Na 5.27%, O 36.66%, S 7.35%.