8952. Sulfisoxazole
Nomenclature
CAS number: 127-69-5
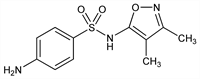
4-Amino-N-(3,4-dimethyl-5-isoxazolyl)benzenesulfonamide; N1-(3,4-dimethyl-5-isoxazolyl)sulfanilamide; 3,4-dimethyl-5-sulfanilamidoisoxazole; 5-(4-aminophenylsulfonamido)-3,4-dimethylisoxazole; 5-(p-aminobenzenesulfonamido)-3,4-dimethylisooxazole; sulfafurazole; sulphafurazole. C11H13N3O3S; mol wt 267.30.
C 49.43%, H 4.90%, N 15.72%, O 17.96%, S 12.00%.
Description and references
Prepn: H. M. Wuest, M. Hoffer, US 2430094 (1947 to Hoffmann-La Roche). Toxicity study: Seki et al., Arzneim.-Forsch. 15, 1441 (1965). Comprehensive description: B. C. Rudy, B. Z. Senkowski, Anal. Profiles Drug Subs. 2, 487-506 (1973). HPLC determn in biological fluids: D. Jung, S. Oie, Clin. Chem. 26, 51 (1980). Clinical trial in otitis media: P. A. M. Bernard et al., Pediatrics 88, 215 (1991).
Properties
White to slightly yellowish crystalline powder. Bitter taste. mp 194°. pKa 5. Soly in water (25°): 0.13 mg/ml. Sol in alcohol. LD50 orally in mice: 6800 mg/kg (Seki).Derivative
Diethanolamine salt.Nomenclature
CAS number: 4299-60-9
Sulfisoxazole diolamine. C11H13N3O3S.C4H11NO2; mol wt 372.44.
C 48.37%, H 6.50%, N 15.04%, O 21.48%, S 8.61%.
Properties
White to off-white, odorless, crystalline powder. Sol in alcohol; freely sol in water. A 4% soln is about isotonic with tears.Derivative
Acetyl sulfisoxazole.Nomenclature
CAS number: 80-74-0
N-[(4-Aminophenyl)sulfonyl]-N-(3,4-dimethyl-5-isoxazolyl)acetamide; N1-monoacetyl sulfisoxazole; Gantrisin (Roche). C13H15N3O4S; mol wt 309.34.
C 50.47%, H 4.89%, N 13.58%, O 20.69%, S 10.37%.
Description and references
Prepn: M. Hoffer, US 2721200 (1955 to Hoffmann-La Roche). Should not be confused with N4-acetyl sulfisoxazole which is a metabolite.
Properties
Tasteless crystals, mp 193-194°. Soly (mg/ml): 0.07 in water; 4.93 in methanol; 5.7 in 95% ethanol; 0.94 in ether; 29.0 in chloroform.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Sulfonamides