9006. Suramin Sodium
Nomenclature
Description and references
Trypanocide discovered in 1917 by O. Dressel and R. Kothe: J. Dressel, J. Chem. Educ. 38, 620 (1961). Prepn: E. Fourneau et al., Compt. Rend. 178, 675 (1924); J. Trefouel, E. Fourneau, GB 224849 (1923); B. Heymann, Angew. Chem. 37, 585 (1924). Pharmacology, toxicology, and efficacy in onchocerciasis and trypanosomiasis: F. Hawking, Adv. Pharmacol. Chemother. 15, 289-322 (1978). Inhibition of reverse transcriptase: E. De Clercq, Cancer Lett. 8, 9 (1979); of infectivity of HIV: H. Mitsuya et al., Science 226, 172 (1984). HPLC determn in plasma: R. W. Klecker, J. M. Collins, J. Liq. Chromatogr. 8, 1685 (1985). Review of biological activities and clinical experience in infection and carcinoma: T. E. Voogd et al., Pharmacol. Rev. 45, 177-203 (1993); C. A. Stein, Cancer Res. 53, 2239-2248 (1993).
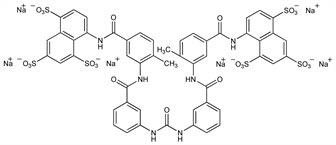
Properties
White or slightly pink or cream-colored powder. Slightly bitter taste. Hygroscopic. Freely sol in water, in physiological saline; sparingly sol in 95% alcohol. Insol in benzene, ether, petr ether, chloroform. Aq solns are neutral to litmus. LD50 in mice (mg/kg): ≈620 i.v. (Hawking).Therapeutic Category
Anthelmintic (Nematodes); antiprotozoal (Trypanosoma).
Therapeutic Category (Veterinary)
Antiprotozoal (Trypanosoma).Keywords
Anthelmintic (Nematodes); Antiprotozoal (Trypanosoma)