9010. Swertiamarin
Nomenclature
CAS number: 17388-39-5
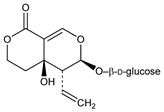
[4aR-(4aα,5β,6α)]-5-Ethenyl-6-(β-d-glucopyranosyloxy)-4,4a,5,6-tetrahydro-4a-hydroxy-1H,3H-pyrano[3,4-c]pyran-1-one; 4,4a,5,6-tetrahydro-4aα-hydroxy-1-oxo-5β-vinyl-1H,3H-pyrano[3,4-c]pyran-6-yl β-d-glucopyranoside. C16H22O10; mol wt 374.34.
C 51.34%, H 5.92%, O 42.74%.
Description and references
Bitter principle of Swertia japonica (Maxim.) Makino, Gentianaceae. Yields erythrocentaurin on hydrolysis with emulsin. Isoln: Kubota, Tomita, Chem. Ind. (London) 1958, 229; Inouye et al., Chem. Pharm. Bull. 18, 1856 (1970). Occurrence in gentianaceous plants: Inouye, Nakamura, J. Pharm. Soc. Jpn. 91, 755 (1971), C.A. 75, 95431b (1971). Structure: Kubota, Tomita, Tetrahedron Lett. 1961, 176; Bull. Chem. Soc. Jpn. 34, 1345 (1961); Koch et al., Bull. Soc. Chim. Fr. 1964, 405. Stereochemistry: Inouye et al., Tetrahedron Lett. 1968, 4429. Biosynthesis studies: Inouye et al., Chem. Pharm. Bull. 18, 2043 (1970).
Properties
Plates from ethanol + chloroform + ether, mp 113-114°. [α]D20 127° (c = 1 in 96% ethanol). uv max (methanol): 238 nm (log ε 3.93).Derivative
Tetraacetate.C24H30O14; mol wt 542.49.
C 53.14%, H 5.57%, O 41.29%.