9048. Tamoxifen
Nomenclature
CAS number: 10540-29-1
(Z)-2-[4-(1,2-Diphenyl-1-butenyl)phenoxy]-N,N-dimethylethanamine; 1-p-β-dimethylaminoethoxyphenyl-trans-1,2-diphenylbut-1-ene. C26H29NO; mol wt 371.51.
C 84.06%, H 7.87%, N 3.77%, O 4.31%.
Description and references
Nonsteroidal estrogen antagonist. Prepn: BE 637389 (1964 to ICI). Identification and separation of isomers: G. R. Bedford, D. N. Richardson, Nature 212, 733 (1966); BE 678807; M. J. K. Harper et al., US 4536516 (1966, 1985 both to ICI). Stereospecific synthesis: R. B. Miller, M. I. Al-Hassan, J. Org. Chem. 50, 2121 (1985). Review of chemistry and pharmacology: B. J. A. Furr, V. C. Jordan, Pharmacol. Ther. 25, 127-205 (1984). Reviews of clinical experience in treatment and prevention of breast cancer: I. A. Jaiyesimi et al., J. Clin. Oncol. 13, 513-529 (1995); C. K. Osborne, N. Engl. J. Med. 339, 1609-1618 (1998).
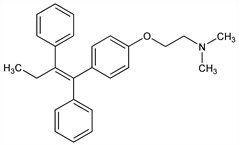
Properties
Crystals from petr ether, mp 96-98°.Derivative
Citrate.Nomenclature
CAS number: 54965-24-1
ICI-46474; Kessar (Pharmacia); Nolvadex (AstraZeneca); Tamofène (Aventis); Zemide (Alpharma); Zitazonium (Servier). C26H29NO.C6H8O7; mol wt 563.64.
C 68.19%, H 6.62%, N 2.49%, O 22.71%.
Properties
Fine, white, odorless crystalline powder, mp 140-142°. Slightly sol in water; sol in ethanol, methanol, acetone. Hygroscopic at high relative humidities. Sensitive to uv light. LD50 in mice, rats (mg/kg): 200, 600 i.p.; 62.5, 62.5 i.v.; 3000-6000, 1200-2500 orally (Furr, Jordan).Derivative
(E)-Form.Nomenclature
CAS number: 13002-65-8
Properties
mp 72-74° from methanol.Derivative
(E)-Form citrate.Nomenclature
ICI-47699.Properties
mp 126-128°.Caution
Tamoxifen is listed as a known human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-239.Therapeutic Category
Antineoplastic (hormonal).
Keywords
Antineoplastic (Hormonal); Antiestrogens; Selective Estrogen Receptor Modulator (SERM)