9052. Tannic Acid
Nomenclature
Description and references
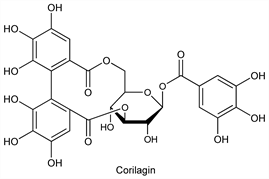
Incorrectly “digallic acid”. Tannic acid of commerce usually contains about 10% H2O. Occurs in the bark and fruit of many plants, notably in the bark of the oak species, in sumac and myrobalan. It is produced from Turkish or Chinese nutgall, the former contg 50-60%, the latter about 70%. The chemistry of the tannins is most complex and non-uniform. Tannins may be divided into 2 groups: (a) derivatives of flavanols, so-called condensed tannins and (b) hydrolyzable tannins (the more important group) which are esters of a sugar, usually glucose, with one or more trihydroxybenzenecarboxylic acids. The structure given here is that of a tannin named corilagin: Schmidt et al., Ann. 587, 67 (1954). The empirical formula of corilagin is C27H22O18 . For the commercial tannic acid, whose specifications follow, the empirical formula is usually given as C76H52O46 . Toxicity study: Robinson, Graessle, J. Pharmacol. Exp. Ther. 77, 63 (1943). Comprehensive reviews: M. Nierenstein, The Natural Organic Tannins (London, 1934); O. Th. Schmidt, “Gallotannine” in Fortschr. Chem. Org. Naturst. 13, 70-136 (1956); Symposium on the Chemistry of Vegetable Tannins (Soc. Leather Trades Chemists, Croydon 1956).
Properties
Yellowish-white to light brown, amorphous, bulky powder or flakes, or spongy masses; faint characteristic odor; astringent taste. Gradually darkens on exposure to air and light; at 210-215° dec mostly into pyrogallol and CO2. Gives insol ppts with albumin, starch, gelatin, most alkaloidal and metallic salts; produces a bluish-black color or precip with ferric salts. One gram dissolves in 0.35 ml water, 1 ml warm glycerol; very sol in alc, acetone. Practically insol in benzene, chloroform, ether, petr ether, carbon disulfide, carbon tetrachloride. Keep well closed and protected from light. LD100 orally in mice: 6.0 g/kg (Robinson, Graessle).Use
Mordant in dyeing; manuf ink; sizing paper and silk; printing fabrics; with gelatin and albumin for manuf of imitation horn and tortoise shell; tanning; clarifying beer or wine; in photography; as coagulant in rubber manuf; manuf gallic acid and pyrogallol; as reagent in analytical chemistry for pH control.Therapeutic Category
Astringent.
Therapeutic Category (Veterinary)
Astringent, hemostatic, in solutions for burns. Has been used internally as an astringent and as a heavy metal antidote.Keywords
Astringent