9062. Taraxerol
Nomenclature
CAS number: 127-22-0
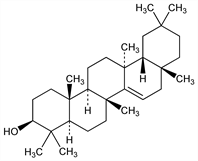
(3β)-d-Friedoolean-14-en-3-ol; isoolean-14-en-3β-ol; skimmiol; alnulin; tiliadin. C30H50O; mol wt 426.72.
C 84.44%, H 11.81%, O 3.75%.
Description and references
Found in Tilia cordata Mill., Tiliaceae: Brutigam, Arch. Pharm. 238, 555 (1900); in Alnus glutinosa (L.) Gaertn., Betulaceae: Zellner, Weiss, Sitzungsber. Akad. Wiss. Wien 132, 258 (1923); in Taraxacum officinale Weber, Compositae: Burrows, Simpson, J. Chem. Soc. 1938, 2042; in Litsea dealbata Nees, Lauraceae: Dunstan et al., Aust. J. Chem. 6, 321 (1953); from Befaria racemosa (Vent.), Ericaceae: Euda et al., J. Org. Chem. 26, 271 (1961). Structure: Beaton et al., J. Chem. Soc. 1955, 2131. Partial synthesis from β-amyrin: eidem, Chem. Ind. (London) 1955, 35.
Properties
Plates from chloroform + methanol, needles from benzene, mp 282-285°. uv max (ethanol): 210, 215, 220, 223 nm (ε 3900, 2400, 700, 250). Sol in benzene, chloroform, ether, ethyl acetate, acetic anhydride, acetic acid, phenol, pyridine, xylene; less sol in alcohol.Derivative
Acetate.C32H52O2; mol wt 468.75.
C 81.99%, H 11.18%, O 6.83%.
Properties
Plates from chloroform + methanol, mp 303-305°. [α]D +10.5° (c = 1.8 in chloroform).Derivative
Benzoate.C37H54O2; mol wt 530.82.
C 83.72%, H 10.25%, O 6.03%.