9118. Teleocidins
Nomenclature
Description and references
Indole alkaloid tumor promoters isolated from the mycelia of Streptomyces. Consists of teleocidin A (two isomers) and, the major component, teleocidin B (four isomers). The mixture is known as telocidin; the name is derived from its toxicity to fish (Teleostei). Isoln: M. Takashima, H. Sakai, Bull. Agric. Chem. Soc. Jpn. 24, 647 (1960). Resolution into A and B and their isomers: H. Fujiki, T. Sugimura, Cancer Surv. 2, 539 (1983). Structure: S. Sakai et al., Chem. Pharm. Bull. 32, 354 (1984). Abs config: Y. Endo et al., ibid. 358. Structure determn of B isomers: Y. Hitotsuyanagi et al., ibid. 4233. Structure-activity study: T. Kawai et al., J. Med. Chem. 35, 2248 (1992). Tumor promotion: H. Fujiki et al., Biochem. Biophys. Res. Commun. 90, 976 (1979); eidem, Carcinogenesis 3, 895 (1982). Review: H. Fujiki, T. Sugimura, Adv. Cancer Res. 49, 233-264 (1987).
Properties
White-crystal like powder, dec >61°. Easily sol in methanol, ethanol, ether, acetone, ethyl-acetate, benzene, chloroform, carbon tetrachloride; slightly sol in petroleum-ether, petroleum-benzine, ligroin. Almost insol in water, %5 HCl and 5% NaOH solution. LD50 orally in mice: 2 mg/kg (Takashima).Derivative
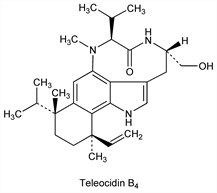
Teleocidin B4.Nomenclature
Derivative
Dihydroteleocidin B.Description and references
Structure: H. Harada et al., Bull. Chem. Soc. Jpn. 39, 1773 (1966). Tumor promoting activity: H. Fujiki et al., Proc. Natl. Acad. Sci. USA 78, 3872 (1981).