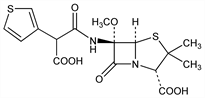
9137. Temocillin
Nomenclature
Description and references
Semi-synthetic injectable penicillin deriv with high activity vs a large number of gram-negative bacteria, but with little activity vs gram-positive organisms. Prepn: J. P. Clayton, P. H. Bentley, DE 2600866 (1976 to Beecham), C.A. 86, 55420t (1977); P. H. Bentley et al., J. Chem. Soc. Perkin Trans. 1 1979, 2455. In vitro antibacterial activity and β-lactamase susceptibility: I. Phillips et al., J. Antimicrob. Chemother. 10, 271 (1982). Comparison to other penicillins vs H. influenzae and intestinal gram-negative rods: H. Y. Chen, J. D. Williams, ibid. 279. Pharmacokinetics and tissue penetration in healthy volunteers: R. M. Brown et al., ibid. 295. In vivo and in vitro comparison to ampicillin: R. Yogev et al., Antimicrob. Agents Chemother. 23, 182 (1983). Series of articles on microbiology, pharmacology and clinical studies: Drugs 29, Suppl. 5, 1-243 (1985).
Derivative
Disodium salt.Nomenclature
Properties
Amorphous solid.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins