9163. Terfenadine
Nomenclature
CAS number: 50679-08-8
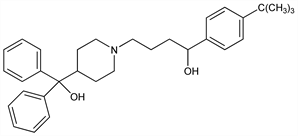
α-[4-(1,1-Dimethylethyl)phenyl]-4-(hydroxydiphenylmethyl)-1-piperidinebutanol; 1-(p-tert-butylphenyl)-4-[4′-(α-hydroxydiphenylmethyl)-1′-piperidyl]butanol; α-(p-tert-butylphenyl)-4-(α-hydroxy-α-phenylbenzyl)-1-piperidinebutanol; MDL-9918; Allerplus (AstraZeneca); Cyater (Lederle); Seldane (Aventis); Teldane (Aventis); Teldanex (AstraZeneca); Terfex (Leiras); Ternadin (Cantabria); Triludan (Aventis). C32H41NO2; mol wt 471.67.
C 81.49%, H 8.76%, N 2.97%, O 6.78%.
Description and references
Nonsedating-type histamine H1-receptor antagonist. Prepn: A. A. Carr, C. R. Kinsolving, DE 2303306; eidem, US 3878217 (1973, 1975 both to Richardson-Merrell); A. A. Carr, D. R. Meyer, Arzneim.-Forsch. 32, 1157 (1982). Series of articles on chemistry, pharmacology, toxicology and clinical studies: ibid. 32, 1153-1218 (1982). Metabolism in human liver: M. Jurima-Romet et al., Drug Metab. Dispos. 22, 849 (1994). Comprehensive description: A. A. Badwan et al., Anal. Profiles Drug Subs. 19, 627-662 (1990). Review: H. C. Masheter, Clin. Rev. Allergy 11, 5-34 (1993).
Properties
Crystals from acetone, mp 146.5-148.5°; may exist in three polymorphic forms, mp 149-152°, 146-148°, 142-144°. Soly at 30° (g/100ml): water 0.001; ethanol 3.780; methanol 3.750; hexane 0.034; 0.1M HCl 0.012; 0.1M citric acid 0.110; 0.1M tartaric acid 0.045. uv max (methanol): 260 nm (A 660.4); (ethanol): 260 nm (A 671.4); (dichloromethane): 260 nm (A 762.2). LD50 orally in mice: >2000 mg/kg (Carr, Meyer).Therapeutic Category
Antihistaminic.
Keywords
Antihistaminic