9182. Tetrabenazine
Nomenclature
CAS number: 58-46-8
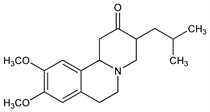
1,3,4,6,7,11b-Hexahydro-9,10-dimethoxy-3-(2-methylpropyl)-2H-benzo[a]quinolizin-2-one; 2-oxo-3-isobutyl-9,10-dimethoxy-1,2,3,4,6,7-hexahydro-11bH-benzo[a]quinolizine; Ro-1-9569. C19H27NO3; mol wt 317.42.
C 71.89%, H 8.57%, N 4.41%, O 15.12%.
Description and references
Dopamine depleting agent. Prepn: Brossi et al., Helv. Chim. Acta 41, 119 (1958); US 2830993 (1958 to Hoffmann-La Roche); Osbond, J. Chem. Soc. 1961, 4711. Pharmacology: Leusen et al., Arch. Int. Pharmacodyn. 119, 225 (1959). Metabolism: Schwartz et al., Biochem. Pharmacol. 15, 645 (1966). Clinical trials in hyperkinetic movement disorders: J. Janovic, Ann. Neurol. 11, 41 (1982); J. Janovic, J. Orman, Neurology 38, 391 (1988).
Properties
Prisms from methanol, mp 125-126°.Derivative
Oxime.Properties
Crystals from dil alcohol, mp 158°.Derivative
Hydrochloride.Nomenclature
CAS number: 2105-47-7
C19H27NO3.HCl; mol wt 353.88.
C 64.49%, H 7.98%, N 3.96%, O 13.56%, Cl 10.02%.
Properties
Crystals, mp 208-210°. Soluble in hot water. Practically insol in acetone. uv max (alcohol): 230, 284 nm (ε 7780, 3820).Derivative
Methanesulfonate.Nomenclature
CAS number: 804-53-5
Nitoman (Roche). C19H27NO3.CH3SO3H; mol wt 413.53.
C 58.09%, H 7.56%, N 3.39%, O 23.21%, S 7.75%.
Properties
Bitter crystals, sensitive to light, mp 126-130°. Sparingly sol in water, sol in alcohol. Practically insol in ether.Therapeutic Category
Antidyskinetic; antipsychotic.
Keywords
Antidyskinetic; Antipsychotic; Other Tricyclics