9196. Tetracycline
Nomenclature
Description and references
Antibiotic substance produced by Streptomyces spp. Prepn: J. H. Boothe et al., J. Am. Chem. Soc. 75, 4621 (1953); L. H. Conover et al., ibid. 4622; Conover, US 2699054 (1955). Production by Streptomyces viridifaciens: Gourevitch, Lein, Heinemann et al., US 2712517; US 2886595 (1955, 1959 both to Bristol Labs.); by S. aureofaciens: Miller, Arishima, Sekizwa, US 3005023; US 3019173 (1961, 1962 both to Am. Cyanamid). Purification: Kaplan, Granatek, US 3301899 (1967 to Bristol-Myers). Total synthesis of tetracyclines: J. H. Boothe et al., J. Am. Chem. Soc. 81, 1006 (1959); L. H. Conover et al., ibid. 84, 3222 (1962). Graphic outline of Woodward synthesis: Chem. Eng. News 40, 36 (Oct. 8, 1962). Abs config: V. N. Dobrynin et al., Tetrahedron Lett. 1962, 901. Solubility studies: Weiss et al., Antibiot. Chemother. 7, 374 (1957). Toxicity: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Review: “Tetracycline” in The Technology of the Tetracyclines vol. I, R. C. Evans, Ed. (Quadrangle Press, New York, 1968) pp 209-426. Mechanism of action: A. Kaji, M. Ryoji in Antibiotics vol. 5 (pt. 1), F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 304-328. Review of biosynthesis: C. R. Hutchinson in Antibiotics vol. 4, J. W. Corcoran, Ed. (Springer-Verlag, New York, 1981) pp 1-12. Review of anticollagenase activity of tetracyclines: L. M. Golub et al., Crit. Rev. Oral Biol. Med. 2, 297-322 (1991).
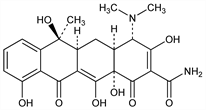
Properties
Trihydrate, crystals. Swells at 165°. Dec 170-175°. Becomes anhydr by drying in vacuo at 60° for 8 hrs. [α]D25 -257.9° (0.1N HCl); [α]D25 -239° (methanol). uv max (0.1N HCl): 220, 268, 355 nm (ε 13000, 18040, 13320). pKa (50% aq DMF): 8.3, 10.2. Stable in neutral and in alkaline soln. Soly at about 28°: 1.7 mg/ml water; >20 mg/ml methanol. LD50 in rats, mice (mg/kg): 807, 808 orally (Goldenthal).Derivative
Hydrochloride.Nomenclature
Properties
Crystals from butanol + HCl, dec 214°. [α]D25 -257.9° (c = 0.5 in 0.1N HCl). Freely sol in water, sol in methanol, ethanol. Insol in ether, hydrocarbons. pH (2% aq soln): 2.1-2.3. LD50 orally in rats: 6443 mg/kg (Goldenthal).Derivative
Phosphate complex.Nomenclature
Description and references
Prepn: Seiger, Weidenheimer, US 3053892 (1962 to Am. Cyanamid).
Properties
Yellow, odorless powder. Sparingly sol in water; slightly sol in ethanol.Therapeutic Category
Antiamebic; antibacterial; antirickettsial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antiamebic; Antibacterial (Antibiotics); Tetracyclines; Antirickettsial