9209. Tetrahydrocannabinols
Description and references
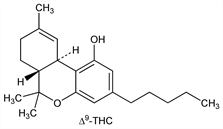
Psychoactive constituents of cannabis, q.v. (Cannabis sativa L. Cannabinaceae). The (-)-trans-Δ9 isomer (Δ9-THC) is the principal active constituent; the (-)-trans-Δ8 isomer (Δ8-THC), although physiologically active, is present to a lesser extent. Δ9 analogs with shorter alkyl side chains have also been identified. Isoln from cannabis resin: H. J. Wollner et al., J. Am. Chem. Soc. 64, 26 (1942). Isoln and structure of Δ9-THC: Y. Gaoni, R. Mechoulam, ibid. 86, 1646 (1964). Isoln of Δ8-THC: R. L. Hively et al., ibid. 88, 1832 (1966). Abs config of (-)-trans-Δ9 isomer: R. Mechoulam, Y. Gaoni, Tetrahedron Lett. 1967, 1109. Stereospecific synthesis of Δ8- and Δ9-THC: R. Mechoulam et al., J. Am. Chem. Soc. 89, 4552 (1967); of Δ9-THC: R. K. Razden et al., ibid. 96, 5860 (1974); eidem, Experientia 31, 16 (1975). IR, NMR, MS data: T. Petrzilka, C. Sikemeier, Helv. Chim. Acta 50, 1416, 2111 (1967). Toxicity studies: H. Rosenkranz et al., Toxicol. Appl. Pharmacol. 28, 18 (1974); H. Yoshimura et al., J. Med. Chem. 21, 1079 (1978). Clinical experience with Δ9-THC in chemotherapy induced nausea and vomiting: D. S. Poster et al., J. Am. Med. Assoc. 245, 2047 (1981); as appetite stimulant in AIDS-associated anorexia: J. E. Beal et al., J. Pain Symptom Manage. 14, 7 (1997). Chromatographic determn in cannabis products: L. Vollner et al., Regul. Toxicol. Pharmacol. 6, 348-358 (1986). GC/MS determn in hair: M. J. Baptista et al., Forensic Sci. Int. 128, 66 (2002). Review of pharmacology: W. L. Dewey, Pharmacol. Rev. 38, 151-178 (1986); of pharmacokinetics and metabolism: F. Grotenhermen, Clin. Pharmacokinet. 42, 327-360 (2003).
Derivative
(-)-trans-Δ9-Form.Nomenclature
Properties
Light yellow resinous oil; hardens upon refrigeration. bp0.02 200°. [α]D20 -150.5° (c = 0.53 in CHCl3). pKa 10.6. uv max (ethanol): 283, 276 nm (log ε 3.21, 3.20). Essentially insol in water. LD50 (sesame oil emulsion) in male rats (mg/kg): 800 orally; 35.5. i.v.; 672 i.p. (Rosenkranz).Derivative
(-)-trans-Δ8-Form.Nomenclature
Properties
bp0.001 200°. [α]D18 -264° (c = 0.11 in ethanol). uv max (ethanol): 282, 275 nm (log ε 3.22, 3.22); shoulder at 230 nm (log ε 4.07). LD50 i.v. in mice: 27.5 mg/kg (Yoshimura).Note
This is a controlled substance (hallucinogen): 21 CFR, 1308.11.Therapeutic Category
Antiemetic; appetite stimulant.
Keywords
Antiemetic