Nomenclature
CAS number: 109-99-9
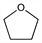
Diethylene oxide; tetramethylene oxide.
C
4H
8O; mol wt 72.11.
C 66.62%, H 11.18%, O 22.19%.
Description and references
Prepn from 1,4-butanediol: Schmoyer, Case, Nature 187, 592 (1960). Manuf
by catalytic hydrogenation of maleic anhydride: Gilbert, Howk, US 2772293 (1956 to du Pont); of furan: Banford, Manes, US 2846449 (1958 to du Pont); Manly, US 3021342 (1962 to Quaker Oats). Stabilization to prevent excessive peroxide formation on
storage with 0.05-1.0% p-cresol, 0.05-0.1% hydroquinone,
or less than 0.01-0.1% 4,4′-thiobis(6-tert-butyl-m-cresol): Bordner, Hinegardner, US 2489260; US 2525410; Campbell, US 3029257 (1949, 1950, 1962 all to du Pont). Review of toxicology and
biological effects: D. E. Moody, Drug Chem.
Toxicol. 14, 319-342 (1991).
Properties
Liquid. Ether-like odor. Flammable. mp -108.5°. d420 0.8892. bp760 66°; bp176 25°. Flash pt 1°F. nD20 1.4070. Dipole moment: 1.70. uv cut-off for spectro grade: 220
nm. Miscible with water, alcohols, ketones, esters, ethers, and hydrocarbons.
Caution: Distil only in presence of a reducing agent,
such as ferrous sulfate; peroxide explosions have occurred: Angew. Chem. 68, 182 (1956).Caution
Potential symptoms of overexposure
are irritation of eyes and upper respiratory system; nausea, dizziness
and headache; CNS depression. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140,
1997) p 302.Use
Solvent for high polymers, esp polyvinyl chloride.
As reaction medium for Grignard and metal hydride reactions. In
the synthesis of butyrolactone, succinic acid, 1,4-butanediol diacetate.
Solvent in histological techniques. May be used under Federal Food,
Drug & Cosmetic Act for fabrication of articles for packaging,
transporting, or storing of foods if residual amount does not exceed
1.5% of the film:
Fed. Regist. 27, 3919 (Apr. 25, 1962).
