Nomenclature
CAS number: 509-14-8
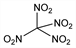
CN
4O
8; mol wt 196.03.
C 6.13%, N 28.58%, O 65.29%.
Description and references
Prepd by nitration of acetic anhydride with
anhydrous nitric acid: Liang, Org. Synth. coll. vol. III, p 803 (1955). Energy of decompn: Tschinkel, Ind. Eng. Chem. 48, 732 (1956).
Properties
Pale yellow liquid. Oxidizer;
poisonous. d425 1.6229; d425 1.638 (tech). mp 13.8°. bp760 126°; bp25.8 40°; bp14.9 30°; bp8.4 20°; bp5.7 13.8°; bp1.9 0°. nD20 1.4384; nD25 1.4358. Viscosity at 20° = 1.76 cP.
Insol in water. Freely sol in alcohol, ether, alcoholic KOH. Attacks
iron, copper, brass, zinc, rubber. Highly explosive in the presence
of impurities.Caution
Potential symptoms of overexposure
are irritation of eyes, skin, nose and throat; dizziness, headache;
chest pain, dyspnea; methemoglobinuria, cyanosis; skin burns. See NIOSH Pocket Guide to Chemical
Hazards (DHHS/NIOSH 97-140, 1997) p 304. See also Patty's Industrial Hygiene and Toxicology vol. 2A, G. D. Clayton, F. E. Clayton, Eds. (John Wiley
& Sons, New York, 4th ed., 1994) 614-617. This substance is reasonably
anticipated to be a human carcinogen: Report
on Carcinogens, Eleventh Edition (PB2005-104914, 2004)
p III-246.Use
Oxidizer in rocket propellants. As explosive in
admixture with toluene. To increase cetane number of diesel fuels.
Reagent for detecting the presence of double bonds in organic compds
and for mild nitrations.
