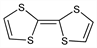
9242. Tetrathiafulvalene
Nomenclature
Description and references
Of interest in solid state chemistry as a conductor, catalyst or sensor due to its unusual electronic and magnetic properties. Primarily used as the parent compound for supramolecular assemblies. Prepn: F. Wudl et al., Chem. Commun. 1970, 1453. In combination with TCNQ (tetracyano-p-quinodimethane) to form first “organic metal”: J. Ferraris et al., J. Am. Chem. Soc. 95, 948 (1973); electronic structure: R. Gleiter et al., J. Electron. Spectrosc. Relat. Phenom. 2, 207 (1973). Use as a catalyst in radical reactions: R. J. Fletcher et al., J. Chem. Soc. Perkin Trans. 1 1995, 623; J. A. Murphy, S. J. Roome, ibid. 1349. TTF-mediated biosensors for glucose: T. Yu et al., J. Appl. Polym. Sci. 58, 973 (1995); for NADH: X. Zhang et al., Anal. Commun. 33, 111 (1996); for gln and glu: A. Mulchandani, A. S. Bassi, Biosens. Bioelectron. 11, 271 (1996). Review: T. Jorgensen et al., Chem. Soc. Rev. 23, 41-51 (1994). Review of electrical properties and structural data of unsymmetric TTF cmpds: J. M. Fabre et al., Synth. Met. 35, 57-64 (1990); of role in superconductors: M. R. Bryce, J. Mater. Chem. 5, 1481-1496 (1995).