9250. Tetroxoprim
Nomenclature
CAS number: 53808-87-0
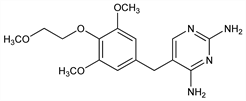
5-[[3,5-Dimethoxy-4-(2-methoxyethoxy)phenyl]methyl]-2,4-pyrimidinediamine; 2,4-diamino-5-[3,5-dimethoxy-4-(2-methoxyethoxy)benzyl]pyrimidine; HE-781. C16H22N4O4; mol wt 334.37.
C 57.47%, H 6.63%, N 16.76%, O 19.14%.
Description and references
Analog of trimethoprim, q.v. Prepn: W. Liebenow, J. Prikryl, FR 2221147; eidem, US 3992379 (1974, 1976 both to Heumann). Series of articles on tetroxoprim and other antibacterial folate inhibitors: J. Antimicrob. Chemother. 5, Suppl. B, 1-239 (1979); on synthesis of radioactive tetroxoprim, tissue distribution, kinetics, HPLC determn: Arzneim.-Forsch. 30, 307-319 (1980). Effect on bacterial growth kinetics: J. K. Seydel, E. Wempe, Chemotherapy 26, 361 (1980). In vitro activity of the tetroxoprim-sulfadiazine combination: H. Hahn, A. Kirov, Arzneim.-Forsch. 30, 1047 (1980).
Properties
Crystals from water, mp 153-156° (Liebenow, Prikryl); also reported as mp 160.1° (eidem, J. Antimicrob. Chemother. 5, Suppl B, 15 (1979)). Soly at 30° (mg/ml): water 2.65; chloroform 69; n-octanol 1.61. pKb 8.25. LD50 orally in rats: 1357 mg/kg (Liebenow, Prikryl).Derivative
Mixture with sulfadiazine.Nomenclature
CAS number: 73173-12-3
Co-tetroxazine; Biroxin (Roche); Sterinor (Heumann); Tibirox (Roche); Troximin (Galenica). Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); 2,4-Diaminopyrimidines