9290. Thiacetazone
Nomenclature
CAS number: 104-06-3
N-[4-[[(Aminothioxomethyl)hydrazono]methyl]phenyl]acetamide; 2-[[4-(acetylamino)phenyl]methylene]hydrazinecarbothioamide; 4′-formylacetanilide thiosemicarbazone; p-acetamidobenzaldehyde thiosemicarbazone; p-acetylaminobenzaldehyde thiosemicarbazone; p-acetaminobenzylidenethiosemicarbazone; amithiozone; thibone; thioacetazone; Tb I-698; Conteben (Bayer); Livazone (Specia); Myrizone (Squibb); Neustab (Boots); Panrone (Panray); Seroden (Allen & Hanburys); Tebethion; Thiocarbazil; Thioparamizone (Smith & Nephew); Tibione (Schenley); Tiobicina. C10H12N4OS; mol wt 236.29.
C 50.83%, H 5.12%, N 23.71%, O 6.77%, S 13.57%.
Description and references
Prepn: G. Domagk et al., Naturwissenschaften 33, 315 (1946); Behnisch et al., Angew. Chem. 60A, 113 (1948); Chabrier, Cattelain, Bull. Soc. Chim. Fr. 1950M, 52; Das, Mukherjee, J. Am. Chem. Soc. 75, 1241 (1953). Toxicity data: Bavin et al., J. Pharm. Pharmacol. 2, 764 (1950). HPLC determn in body fluids: P. J. Jenner, J. Chromatogr. 276, 463 (1983). Review of clinical experience: H. C. Hinshaw, W. McDermott, Am. Rev. Tuberc. 61, 145-157 (1950); of clinical safety and efficacy: P. Nunn et al., Trans. R. Soc. Trop. Med. Hyg. 87, 578 (1993).
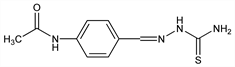
Properties
Minute, pale yellow crystals from abs alc. Bitter taste. Darkens on exposure to light. Dec 225-230°. uv max (ethanol): 328 nm, extinction 0.580. Sol in hot alc; very sparingly sol in cold alc. Insol in water, acetone, benzene, carbon tetrachloride, chloroform, carbon disulfide, petr ether. Practically insol in the other common organic solvents except glycols. Soly in propylene glycol ≈1%. LD50 s.c. in mice: 1-2 g/kg (Bavin).Therapeutic Category
Antibacterial (tuberculostatic).
Keywords
Antibacterial (Tuberculostatic)