9333. Thioformamide
Nomenclature
CAS number: 115-08-2
Methanethioamide. CH3NS; mol wt 61.11.
C 19.65%, H 4.95%, N 22.92%, S 52.47%.
Description and references
Prepd by the action of phosphorus pentasulfide on formamide: Willsttter, Wirth, Ber. 42, 1911 (1909). Synthesis: Erlenmeyer, Menzi, Helv. Chim. Acta 31, 2071 (1948); Schmitz, US 2682558 (1954 to du Pont); R. Tull, L. M. Weinstock, Angew. Chem. Int. Ed. 8, 278 (1969). Spectroscopic studies: R. Sugisaki et al., J. Mol. Spectrosc. 49, 241 (1974); R. H. Judge et al., Can. J. Chem. 65, 2100 (1987). Use in synthesis of thiazoles: T. J. Cousineau, J. A. Secrist III, J. Org. Chem. 44, 4351 (1979); H. Fukatsu et al., Heterocycles 29, 1517 (1989).
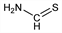
Properties
Prisms from ethyl acetate or from ether + petr ether, mp 26-27°. uv max (CH3OH): 263 nm (ε 12500). Turns yellow and dec to a dark-colored mass. May be stored as soln in abs ether, preferably over P2O5. Sol in water (with considerable cooling). Freely sol in tetrahydrofuran, alcohol, ether, acetone, ethyl acetate; sparingly sol in cold chloroform, benzene, petr ether, carbon disulfide. The decompn products are hydrogen cyanide, hydrogen sulfide, ammonia, and some solid, amorphous, sulfur-contg compounds.Derivative
Monohydrate.Nomenclature
CAS number: 5967-97-5
CH3NS.H2O; mol wt 79.12.
C 15.18%, H 6.37%, N 17.70%, S 40.53%, O 20.22%.