9365. Thiothixene
Nomenclature
CAS number: 5591-45-7
N,N-Dimethyl-9-[3-(4-methyl-1-piperazinyl)propylidene]thioxanthene-2-sulfonamide; cis-9-[3-(4-methyl-1-piperazinyl)propylidene]-2-(dimethylsulfonamido)thioxanthene; tiotixene; Navane (Roerig); Orbinamon (Pfizer). C23H29N3O2S2; mol wt 443.63.
C 62.27%, H 6.59%, N 9.47%, O 7.21%, S 14.46%.
Description and references
Prepn of cis/trans-isomer mixture and sepn of isomers: Bloom, Muren, BE 647066 corresp to US 3310553 (1964, 1967 both to Pfizer); Muren, Bloom, J. Med. Chem. 13, 17 (1970). Only cis-isomer exhibits therapeutic activity. Structure studies: J. P. Schaefer, Chem. Commun. 1967, 743. Pharmacological studies: A. Weissman, Psychopharmacologia 12, 142 (1968). HPLC determn in serum: C. Dilger et al., Arzneim.-Forsch. 38, 1522 (1988). Book: T. A. Ban, Psychopharmacology of Thiothixene (Raven Press, New York, 1978) 485 pp. Comprehensive description: D. K. Wyatt, L. T. Grady, Anal. Profiles Drug Subs. 18, 527-565 (1989).
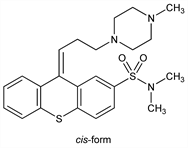
Derivative
cis-Isomer.Properties
Crystals, mp 147.5-149°. uv max (methanol): 228, 260, 310 nm (log ε 4.6, 4.3, 3.9). Has greater pharmacologic activity than trans-isomer. LD50 in mice, rats (mg/kg): 100, 55 i.p. (Weissman).Derivative
trans-Isomer.Properties
mp 123-124.5°. uv max (methanol): 229, 252, 301 nm (log ε 4.5, 4.2, 3.9). LD50 i.p. in mice: 235 mg/kg (Weissman).Derivative
cis-trans-Isomer mixture.Properties
Crystals, mp 114-118°.Derivative
Dimaleate.Properties
mp 158-160.5°.Derivative
Dioxalate.Properties
mp 229°.Therapeutic Category
Antipsychotic.
Keywords
Antipsychotic; Thioxanthenes