Nomenclature
CAS number: 62-56-6
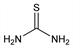
Thiocarbamide.
CH
4N
2S; mol wt 76.12.
C 15.78%, H 5.30%, N 36.80%, S 42.12%.
Description and references
Made by fusing ammonium thiocyanate: Powers
and Powers, Mitchell, US 2552584; US 2560596 (both 1951 to Koppers); by treating cyanamide with
hydrogen sulfide: Robin, Jr., US 2173067 (1940 to Am. Cyanamid); Lewis, US 2393917 (1946 to Monsanto); Van de Kamp, US 2357149 (1944 to Merck & Co.). Toxicity data: Dieke et al., J. Pharmacol. Exp. Ther. 90, 262 (1947).
Properties
Crystals, mp 176-178°. d 1.405. Soluble in 11 parts water, in alcohol; sparingly sol in ether.
Neutral reaction. Forms addition compds with metallic salts. LD50 orally in wild
Norway rats: 1830 mg/kg (Dieke).Caution
Chronic administration in rats has
resulted in hepatic tumors, bone marrow depression and goiters: Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins,
Baltimore, 5th ed., 1984) Section II, p 350. This substance is reasonably
anticipated to be a human carcinogen: Report
on Carcinogens, Eleventh Edition (PB2005-104914, 2004)
p III-250.Use
In animal glue liquifiers and silver tarnish removers.
Photographic fixing agent and to remove stains from negatives; manuf
resins; vulcanization accelerator; a reagent for determn of bismuth,
selenite ions.
