9392. Thujone
Nomenclature
4-Methyl-1-(1-methylethyl)bicyclo[3.1.0]hexan-3-one; 3-thujanone.C10H16O; mol wt 152.23.
C 78.90%, H 10.59%, O 10.51%.
Description and references
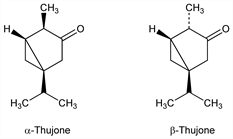
A constituent of many essential oils; present in thuja, etc. Equilibrium mixture contains 33% α-thujone and 67% β-thujone: Eastman, Winn, J. Am. Chem. Soc. 82, 5908 (1960). α- and β-Thujones differ only in the stereochemistry of the 4-methyl group. Conformation: Hach et al., Tetrahedron Lett. 1970, 3175. Chemistry: J. P. Kutney et al., Bioorg. Chem. 7, 289 (1978); eidem, Can. J. Chem. 57, 3145 (1979); 58, 2641 (1980). Toxicity study: K. C. Rice, R. S. Wilson, J. Med. Chem. 19, 1054 (l976). Review: J. L. Simonsen, The Terpenes vol. II (University Press, Cambridge, 1949) pp 32-52.
Properties
Colorless or almost colorless liquid. uv max (isooctane): 300 nm (ε 23). Practically insol in water. Sol in alc and many other organic solvents. LD50 s.c. in mice: 134.2 mg/kg (Rice, Wilson).Derivative
α-Thujone.Nomenclature
CAS number: 546-80-5
(1S,4R,5R)-()-3-Thujanone. Properties
bp17 83.8-84.1°. d425 0.9109. nD15 1.4490. [α]D20 19.2°. LD50 s.c. in mice: 87.5 mg/kg (Rice, Wilson).Derivative
β-Thujone.Nomenclature
CAS number: 471-15-8
d-Isothujone; (1S,4S,5R)-(+)-3-thujanone.