9435. Tigogenin
Nomenclature
Description and references
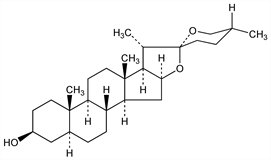
The aglycon of tigonin; C-5 epimer of smilagenin, q.v. Obtained from leaves of Digitalis lanata Ehrh., Scrophulariaceae: Windhaus, Z. Physiol. Chem. 150, 205 (1925); Jacobs, Fleck, J. Biol. Chem. 88, 545 (1930). From the sisal plant Agave sisalana L., Amaryllidaceae: Rubin, US 2991282 (1961); US 3303187 (1967). Structure: Marker, Rohrmann, J. Am. Chem. Soc. 62, 898 (1940); and synthesis: Mazur, Sondheimer, ibid. 81, 3161 (1959); Caglioti, Magi, Tetrahedron 19, 1127 (1963). In synthesis of steroids: T. Ohta et al., Org. Process Res. Dev. 1, 420 (1997); L. Amiranshvili et al., Bull. Georgian Acad. Sci. 161, 252 (2000).
Properties
Crystals from dil methanol, mp 203°. [α]D20 62°. More sol in acetone, in ether, and esp in petr ether than gitogenin. Pptd by digitonin.Derivative
Tigonin.Nomenclature
Description and references
Isoln from Digitalis sp: Tschesche, Ber. 69, 1665 (1936); Liang, Noller, J. Am. Chem. Soc. 57, 525 (1935). Built from 2 glucose, 2 galactose, 1 xylose, and 1 tigogenin unit.