9484. Tixocortol
Nomenclature
CAS number: 61951-99-3
(11β)-11,17-Dihydroxy-21-mercaptopregn-4-ene-3,20-dione; 11β,17α-dihydroxy-21-thio-3,20-dioxo-4-pregnene. C21H30O4S; mol wt 378.53.
C 66.63%, H 7.99%, O 16.91%, S 8.47%.
Description and references
Synthesis and biological activity: S. S. Simons et al., J. Steroid Biochem. 13, 311 (1980). Use in fluorescent chemoaffinity labeling: eidem, Biochemistry 18, 4915 (1979). Prepn of the 21-pivalate: D. R. Torossian et al., DE 2357778; eidem, US 4014909 (1974, 1977 both to Jouveinal). Series of articles on pharmacology, in vitro and in vivo activity of the pivalate: Arzneim.-Forsch. 31, 453-469 (1981).
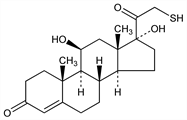
Properties
Fine white solid, dec 220-221°. uv max (95% ethanol): 241 nm (ε 1.65×104).Derivative
21-Pivalate.Nomenclature
CAS number: 55560-96-8
(11β)-21-[(2,2-Dimethyl-1-oxopropyl)thio]-11,17-dihydroxypregn-4-ene-3,20-dione; JO-1016; Pivalone (Jouveinal); Rectovalone (Jouveinal); Tiovalon (Intersan). C26H38O5S; mol wt 462.64.
C 67.50%, H 8.28%, O 17.29%, S 6.93%.
Properties
Crystals from ethanol, mp 195-200°. [α]D20 +145° (c = 1 in dioxane). uv max (methanol): 229 nm (log ε 4.259).Use
Free thiol in chemoaffinity labeling.Therapeutic Category
Anti-inflammatory.
Keywords
Glucocorticoid