9519. Tolnaftate
Nomenclature
CAS number: 2398-96-1
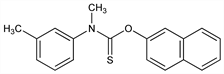
Methyl(3-methylphenyl)carbamothioic acid O-2-naphthalenyl
ester; m,N-dimethylthiocarbanilic acid O-2-naphthyl ester; O-2-naphthyl m,N-dimethylthiocarbanilate; 2-naphthyl N-methyl-N-(3-tolyl)thionocarbamate; Sch-10144; Aftate (Schering-Plough); Chinofungin (Chinoin); Fungistop (Essex); Hi-Alarzin (Yamanouchi); Sporiline (Schering-Plough); Timoped (Reckitt & Colman); Tinactin (Schering-Plough); Tinaderm (Schering-Plough); Tonoftal (Essex). C19H17NOS; mol wt 307.41.
C 74.23%, H 5.57%, N 4.56%, O 5.20%, S 10.43%.
Description and references
Prepn: FR 1337797; Miyazaki et al., US 3334126 (1963, 1967 to Japan Soda); Noguchi et al., J. Pharm. Soc. Jpn. 88, 335 (1968). Pharmacology and toxicology: Noguchi et al., Antimicrob. Agents Chemother. 1962, 259; Hashimoto et al., Toxicol. Appl. Pharmacol. 8, 380 (1966); Noguchi et al., ibid. 368. Clinical comparison with undecylenic acid: J. F. Fuerst et al., Cutis 25, 544 (1980); F. Battistini et al., Int. J. Dermatol. 22, 388 (1983). Mode of action study: M. P. Gupta et al., J. Vet. Med. Mycol. 29, 45 (1991). LC determn in pharmaceuticals: A. K. Dash, J. Pharm. Biomed. Anal. 11, 847 (1993). Comprehensive description: idem, Anal. Profiles Drug Subs. Excip. 23, 543-570 (1994).
Properties
Crystals from alcohol, mp 110.5-111.5°. Practically insol in water. Sparingly sol in methanol, ethanol. Sol in chloroform (1:1.5), acetone (1:7), CCl4 (1:9). uv max (methanol): 258, 222 nm. LD50 in mice, rats (g/kg): >10, >6 orally; >6, >4 s.c. (Hashimoto).Therapeutic Category
Antifungal.
Therapeutic Category (Veterinary)
Antifungal.Keywords
Antifungal (Synthetic); Thiocarbamates