9549. Torcetrapib
Nomenclature
CAS number: 262352-17-0
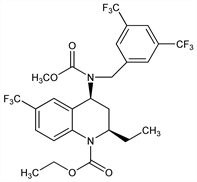
(2R,4S)-4-[[[3,5-Bis(trifluoromethyl)phenyl]methyl](methoxycarbonyl)amino]-2-ethyl-3,4-dihydro-6-(trifluoromethyl)-1(2H)-quinolinecarboxylic acid ethyl ester; (2R,4S)-4-[(3,5-bis-trifluoromethylbenzyl)methoxycarbonylamino]-2-ethyl-6-trifluoromethyl-3,4-dihydro-2H-quinoline-1-carboxylic acid ethyl ester; CP-529414. C26H25F9N2O4; mol wt 600.47.
C 52.01%, H 4.20%, F 28.48%, N 4.67%, O 10.66%.
Description and references
Cholesteryl ester transfer protein (CETP) inhibitor. Prepn: M. P. DeNinno et al., WO 0017164; eidem, US 6197786 (2000, 2001 both to Pfizer); of crystalline forms: D. J. M. Allen et al., WO 0140190 (2001 to Pfizer). Mechanism of action study: R. W. Clark et al., J. Lipid Res. 47, 537 (2006). Clinical evaluation of effects on HDL cholesterol levels: R. W. Clark et al., Arterioscler. Thromb. Vasc. Biol. 24, 490 (2004); M. E. Brousseau et al., N. Engl. J. Med. 350, 1505 (2004). Review of clinical development in combination with atorvastatin: J. R. Burnett, Curr. Opin. Investig. Drugs 6, 944-950 (2005).
Properties
Anhydrous, non-hygroscopic crystals, mp 89-90°. d 1.406.Derivative
Ethanolate.Nomenclature
CAS number: 343798-00-5
C26H25F9N2O4.C2H6O; mol wt 646.54.
C 52.02%, H 4.83%, F 26.45%, N 4.33%, O 12.37%.
Properties
White crystalline powder, mp 54-58°. [α]D 93.3° (c = 1.08 in methanol). d 1.402. Non-hygroscopic. Higher aqueous soly than anhydrous form.Therapeutic Category
Antilipemic; antiatherosclerotic.
Keywords
Antilipemic; CETP Inhibitor; Antiatherosclerotic