9625. Trichlormethine
Nomenclature
CAS number: 555-77-1
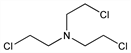
2-Chloro-N,N-bis(2-chloroethyl)ethanamine; 2,2′,2″-trichlorotriethylamine; tris(β-chloroethyl)amine; trimustine; HN-3. C6H12Cl3N; mol wt 204.53.
C 35.23%, H 5.91%, Cl 52.00%, N 6.85%.
Description and references
Nitrogen mustard formerly used as a chemical warfare agent. Prepd by the action of thionyl chloride on triethanolamine: Ward, J. Am. Chem. Soc. 57, 914 (1935); US 2072348 (1937 to Hercules Powder). Improved procedure: Wilson, Tishler, J. Am. Chem. Soc. 73, 3635 (1951); Witten in Kirk-Othmer Encyclopedia of Chemical Technology vol. 7 (Interscience, New York, 1951) p 130. Clinical use in treatment of Hodgkin's disease and leukemias: L. S. Goodman et al., J. Am. Med. Assoc. 132, 126 (1946). Evaluation of carcinogenic risk: IARC Monographs 50, 143 (1990). GC determn in air: J. R. Stuff et al., J. Chromatogr. A 849, 529 (1999).
Properties
Mobile liquid. Faint odor of fish + soap. Vesicant, necrotizing irritant. Never use without appropriate gas mask. Volatility at 25° = 0.120 mg/l. d425 1.2347, mp 4°. bp15 144°. nD25 1.4925. Very slightly sol in water. Miscible with dimethylformamide, carbon disulfide, carbon tetrachloride, many other organic solvents and oils. The undiluted liquid dec on standing and forms polymeric quaternary ammonium salts which are insol in the free base.Derivative
Hydrochloride.Nomenclature
CAS number: 817-09-4
Trillekamin (Crookes). C6H12Cl3N.HCl; mol wt 240.99.
C 29.90%, H 5.44%, Cl 58.85%, N 5.81%.
Properties
Crystals, mp 130-131°. Freely sol in water, sol in alcohol.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Alkylating Agents; Nitrogen Mustards