Nomenclature
CAS number: 118-96-7
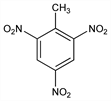
TNT; α-trinitrotoluol;
sym-trinitrotoluene; 1-methyl-2,4,6-trinitrobenzene; trotyl; tolit; trilit.
C
7H
5N
3O
6; mol wt 227.13.
C 37.02%, H 2.22%, N 18.50%, O 42.26%.
Description and references
Prepn by nitration of toluene with mixed acid
(HNO3 + H2SO4) in three steps or
by continuous flow according to the Schmid-Meissner and Biazi processes:
Swift, Tittensor, J. Soc. Chem. Ind. 59, 92 (1940); Johnston in McGraw-Hill
Encyclopedia of Science and Technology 9, 104
(1960). Physical constants and applications: Lothrop, Handrick, Chem. Rev. 44, 419-445 (1949); ACS Monograph Series no. 139,
entitled “The Science of High Explosives,” M. A. Cook, Ed. (Reinhold,
New York, 1958). Review of toxicology and human exposure: Toxicological Profile for 2,4,6-Trinitrotoluene (PB95-264297, 1995) 208 pp.
Properties
Monoclinic rhombohedra from alcohol. The commercial
crystals (needles) are yellow. mp 80.1°. d420 1.654. Burns at 295° when not confined.
Can be distilled under reduced pressure. Vapors are toxic. Dipole
moment 1.37. Very sparingly sol in water: About 0.01% at 25°, one
gram dissolves in 700 ml of boiling water. Sol in acetone, benzene.
Less sol than 2,4,6-trinitrophenol in alcohol, ether, carbon disulfide.
Reacts vigorously with reducing agents.Caution
Potential symptoms of overexposure
are irritation of skin, mucous membranes; liver damage, jaundice;
cyanosis; sneezing; coughing, sore throat; peripheral neuropathy,
muscle pain; kidney damage; cataract; sensitization dermatitis; leukocytosis;
anemia; cardiac irregularities. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140,
1997) p 322.Use
High explosive in military munitions. Must be detonated
by a high velocity initiator such as nitramine or by efficient concussion.
