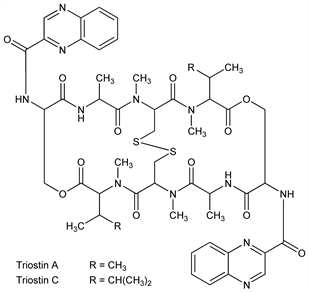
9733. Triostins
Description and references
A “quinoxaline antibiotic complex” similar to echinomycin, q.v. Powerful, selective inhibitor of nucleic acid synthesis in vitro. Isoln of triostin C from Streptomyces S-2-210 resembling S. aureus: J. Shoji, K. Katagiri, J. Antibiot. 14A, 335 (1961). Isoln of triostins A and B: H. Otsuka, J. Shoji, ibid. 19A, 128 (1966). Structure of C: eidem, Tetrahedron 21, 2931 (1965); of minor components: eidem, ibid. 23, 1535 (1967); of A: H. Otsuka et al., J. Antibiot. 29, 107 (1976). Biosynthesis: T. Yoshida, K. Katagiri, Biochemistry 8, 2645 (1969). Synthesis of A: P. K. Chakravarty, R. K. Olsen, Tetrahedron Lett. 1978, 1613; M. Shin et al., Pept. Chem. 18, 207 (1980). Conformation of A in soln: J. R. Kalman et al., J. Chem. Soc. Perkin Trans. 1 1979, 1313. Review of chemistry and biochemistry: M. J. Waring in Antibiotics vol. 5(pt. 2), F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 173-194.