9787. Trovafloxacin
Nomenclature
CAS number: 147059-72-1
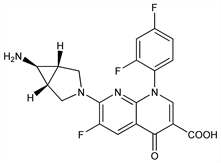
(1α,5α,6α)-7-(6-Amino-3-azabicyclo[3.1.0]hex-3-yl)-1-(2,4-difluorophenyl)-6-fluoro-1,4-dihydro-4-oxo-1,8-naphthyridine-3-carboxylic
acid; CP-99219. C20H15F3N4O3; mol wt 416.35.
C 57.70%, H 3.63%, F 13.69%, N 13.46%, O 11.53%.
Description and references
Fluorinated quinolone antibacterial. Prepn: K. E. Brighty, US 5164402 (1992 to Pfizer). Voltammetric determn in urine and serum: J. L. Vílchez et al., J. Pharm. Biomed. Anal. 31, 465 (2003). Antibacterial spectrum in vitro: H. C. Neu, N.-X. Chin, Antimicrob. Agents Chemother. 38, 2615 (1994). Clinical pharmacokinetics: R. Teng et al., J. Antimicrob. Chemother. 36, 385 (1995). Evaluation in gonorrhea: E. W. Hook et al., Antimicrob. Agents Chemother. 40, 1720 (1996). Mechanism of hepatotoxicity: M. J. Liguori et al., Hepatology 41, 177 (2005).
Derivative
Hydrochloride.Nomenclature
CAS number: 146961-34-4
C20H15F3N4O3.HCl; mol wt 452.81.
C 53.05%, H 3.56%, F 12.59%, N 12.37%, O 10.60%, Cl 7.83%.
Properties
Pale yellow crystals from acetonitrile/methanol, mp 246° (dec).Derivative
Methanesulfonate.Nomenclature
CAS number: 147059-75-4
Trovafloxacin mesylate; CP-99219-27; Trovan. C20H15F3N4O3.CH3SO3H; mol wt 512.46.
C 49.22%, H 3.74%, F 11.12%, N 10.93%, O 18.73%, S 6.26%.
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs