9789. Troxerutin
Nomenclature
CAS number: 7085-55-4
2-[3,4-Bis(2-hydroxyethoxy)phenyl]-3-[[6-O-(6-deoxy-α-l-mannopyranosyl)-β-d-glucopyranosyl]oxy]-5-hydroxy-7-(2-hydroxyethoxy)-4H-1-benzopyran-4-one; 7,3′,4′-tris[O-(2-hydroxyethyl)]rutin; trioxyethylrutin; tri(hydroxyethyl)rutoside; Posorutin (Ursapharm); Ruven (Mepha); Vastribil (Farmasan); Veinamitol (NEGMA); Veniten (Wyeth). C33H42O19; mol wt 742.68.
C 53.37%, H 5.70%, O 40.93%.
Description and references
The principal component of a mixture, the O-(β-hydroxyethyl)rutosides, which also contains mono-, di-, tetra- and other trihydroxyethyl derivs of rutin, q.v. The mixture is prepd by the hydroxyethylation of the phenolic groups of rutin with glycochlorohydrin in alk medium: J. Favre, CH 349614 (1957); see also GB 833174 (1960 to Zyma), C.A. 54, 21135i (1960). Isolation and identification of major components of the mixture: P. Courbat et al., Helv. Chim. Acta 49, 1203, 1420 (1966). Prepn of troxerutin: P. J. Courbat, US 3420815 (1969 to Zyma). Metabolism in man: A. M. Hackett et al., Arzneim.-Forsch. 26, 925 (1976).
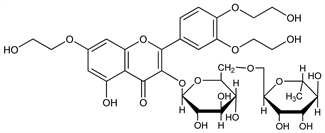
Properties
Yellow powder, mp 181°. Sol in water, glycerol, propylene glycol. Practically insol in cold ethanol, methanol (forms alcoholate), ether, benzene, chloroform.Derivative
O-(β-Hydroxyethyl)rutinosides (mixture).Nomenclature
CAS number: 55965-63-4
HR; Paroven (Zyma); Relvene (Pharmascience); Varemoid (Zyma); Venoruton (Zyma). Properties
Yellow powder, mp 156°. Sol in water, methanol, glycerol, propylene glycol. Practically insol in cold ethanol (forms alcoholate), ether, benzene, chloroform.Therapeutic Category
Treatment of venous disorders.
Keywords
Vasoprotectant