9897. Uzarin
Nomenclature
Description and references
Isoln from the dried root of a Gomphocarpus sp, Asclepiadaceae: Windaus, Haack, Ber. 63, 1377 (1930); Tschesche, Brathge, ibid. 85, 1042 (1952); Schmid et al., Helv. Chim. Acta 42, 72 (1959). Yields uzarigenin by enzymic cleavage. Structure: Tschesche, Bohle, Ber. 68, 2252 (1935). Structure of uzarigenin: Rangaswami, Reichstein, Helv. Chim. Acta 32, 939 (1949); Russel et al., ibid. 44, 1320 (1961). Unlike all other known cardiac glycosides, has the A/B-trans configuration: L. F. Fieser, M. Fieser, Steroids (Reinhold, New York, 1959) pp 762-763. Review: Heusser, Fortschr. Chem. Org. Naturst. 7, 101 (1950).
Properties
Prisms from pyridine + water, mp 266-270°; stout needles from methanol + ether, mp 206-208°. [α]D20 27° (c = 1.075 in pyridine); [α]D19 1.4° (c = 0.85 in methanol). uv max: 217 nm (log ε 4.23). Sol in pyridine, hot methyl Cellosolve; sparingly sol in water. Practically insol in ether, chloroform, acetone.Derivative
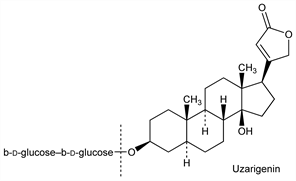
Uzarigenin.Nomenclature
Description and references
Synthesis: Stache et al., Ann. 726, 136 (1969); Kamano et al., J. Org. Chem. 39, 2319 (1974); eidem, J. Chem. Soc. Perkin Trans. 1 1975, 1972.
Properties
Crystals from methanol, mp 240-256°. [α]D20 +10.5° (c = 1.056 in alc).Derivative
3-O-Acetyluzarigenin.Properties
Hexagonal plates from methanol, mp 262-266°. [α]D22 +4.6° (c = 1.09 in chloroform).Therapeutic Category
Antidiarrheal.
Keywords
Antidiarrheal